Poster Session, Thursday, June 17Theme F686 - N1123Fabrication of Plat<strong>in</strong>um Nanoparticles Us<strong>in</strong>g Amphiphilic Copolymer TemplateNuman HODA*, Burç<strong>in</strong> ACAR, Leyla BUDAMA, Önder TOPELAkdeniz University Department of Chemistry Antalya TurkeyAbstract-Production of nanoparticles with controlled size is important for their properties. In this study, production of Ptnanoparticles us<strong>in</strong>g PB-b-PEO amphiphilic copolymer template to control their size by load<strong>in</strong>g different amount of precursor was tried.Synthesis of nanoparticles of metals, semiconductorsand magnetic <strong>crystals</strong> has been accomplished by severalmethods <strong>in</strong>clud<strong>in</strong>g amphiphilic copolymer template. Thesek<strong>in</strong>ds of copolymers have ability to form micelles <strong>in</strong> dilutesolutions <strong>in</strong> selective solvents for one of the blocks [1].Metal salts can be encapsulated <strong>in</strong> the core of micelles bycomplexation or association, corona provides stabilization.Encapsulated metals are easily chemically reduced oroxidized to convert them their nanoparticles.The aim of study is to control Pt nanoparticles size byadd<strong>in</strong>g different amount of precursor to copolymermicelles.PB-b-PEO is a typical amphiphilic copolymer whichcan form micelle <strong>in</strong> aqueous solution. In characterizationof copolymer PB1800-b-PEO4000 (from Polymer Sources,Canada) micelles formed <strong>in</strong> aqueous solution,hydrodynamic diameter of micelles (D h ) was found to be52.4(±2) nm. DLS method also gives <strong>in</strong>formation aboutpolydispersity of the micelles and this was about 0.075.The cmc of PB-b-PEO diblock copolymer <strong>in</strong> aqueoussolution was estimated to be 2.94x10 -7 M by fluorescencespectroscopy.PB-b-PEO diblock copolymer micelles <strong>in</strong> aqueoussolution were used to as nanoreactor to produce Ptnanoparticles hav<strong>in</strong>g certa<strong>in</strong> sizes. To control particle sizeamount of salt to be added was changed. TEM Picture wastaken <strong>in</strong> 120 kV. The PB-b-PEO diblock copolymermicelles from TEM are showed <strong>in</strong> Fig. 1. Result<strong>in</strong>gpictures with different metal:polymer ratio (<strong>in</strong> mol) aregiven <strong>in</strong> Figs. 2.a-f with the size distribution.Figure 1. Micelles of PB-b-PEO <strong>in</strong> aqueous solutionFigure 2. Nanoparticles produced <strong>in</strong> a;(3:1) , b;(1:1), c;(1:5),d;(1:10), e;(1:15), f;(1:20), metal:polymer load<strong>in</strong>g.When metal:polymer ratio is favor of metal for example, 3:1,micelles do not stabilize all of metal salt added accord<strong>in</strong>g toFig.2a. In this figure, there are some agglomerates outside ofthe micelle. It may be understood that the complexationbetween metal salt and hydrophobic part of the copolymer isweak. It can be seen that <strong>in</strong> the other pictures nanoparticlesare dispersed homogeneously. The mean diameters ofnanoparticles for 1:1, 1:5, 1:10 and 1:20 ratios are 1.3(2),1.5(3), 1.4(3) and 1.1(2) nm, respectively. Accord<strong>in</strong>g toTEM pictures of nanoparticles obta<strong>in</strong>ed <strong>in</strong>creas<strong>in</strong>g amount ofmetal salt load<strong>in</strong>g to micelles does not affect much the sizeof nanoparticles. This work was supported by AkdenizUniversity the Scientific Research Projects Coord<strong>in</strong>ationUnit under Grant No. 2007.01.0105.007.*Correspond<strong>in</strong>g Author: nhoda@akdeniz.edu.tr[1] G. Riess, Prog. Polym. Sci., 2003, 28, 1107.6th Nanoscience and Nanotechnology Conference, zmir, 2010 654
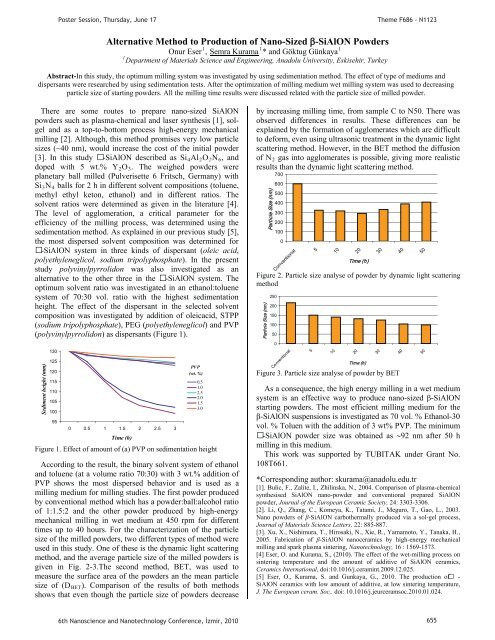
PPoster Session, Thursday, June 17Theme F686 - N1123Alternative Method to Production of Nano-Sized -SiAlON Powders111Onur EserP P, USemra KuramaUP P* and Göktug GünkayaP1PDepartment of Materials Science and Eng<strong>in</strong>eer<strong>in</strong>g, Anadolu University, Eskisehir, TurkeyAbstract-In this study, the optimum mill<strong>in</strong>g system was <strong>in</strong>vestigated by us<strong>in</strong>g sedimentation method. The effect of type of mediums anddispersants were researched by us<strong>in</strong>g sedimentation tests. After the optimization of mill<strong>in</strong>g medium wet mill<strong>in</strong>g system was used to decreas<strong>in</strong>gparticle size of start<strong>in</strong>g powders. All the mill<strong>in</strong>g time results were discussed related with the particle size of milled powder.There are some routes to prepare nano-sized SiAlONpowders such as plasma-chemical and laser synthesis [1], solgeland as a top-to-bottom process high-energy mechanicalmill<strong>in</strong>g [2]. Although, this method promises very low particlesizes (~40 nm), would <strong>in</strong>crease the cost of the <strong>in</strong>itial powder[3]. In this study -SiAlON described as SiR4RAlR2ROR2RNR6R, anddoped with 5 wt.% YR2ROR3R. The weighed powders wereplanetary ball milled (Pulverisette 6 Fritsch, Germany) withSiR3RNR4R balls for 2 h <strong>in</strong> different solvent compositions (toluene,methyl ethyl keton, ethanol) and <strong>in</strong> different ratios. Thesolvent ratios were determ<strong>in</strong>ed as given <strong>in</strong> the literature [4].The level of agglomeration, a critical parameter for theefficiency of the mill<strong>in</strong>g process, was determ<strong>in</strong>ed us<strong>in</strong>g thesedimentation method. As expla<strong>in</strong>ed <strong>in</strong> our previous study [5],the most dispersed solvent composition was determ<strong>in</strong>ed for-SiAlON system <strong>in</strong> three k<strong>in</strong>ds of dispersant (oleic acid,polyethyleneglicol, sodium tripolyphosphate). In the presentstudy polyv<strong>in</strong>ylpyrrolidon was also <strong>in</strong>vestigated as analternative to the other three <strong>in</strong> the -SiAlON system. Theoptimum solvent ratio was <strong>in</strong>vestigated <strong>in</strong> an ethanol:toluenesystem of 70:30 vol. ratio with the highest sedimentationheight. The effect of the dispersant <strong>in</strong> the selected solventcomposition was <strong>in</strong>vestigated by addition of oleicacid, STPP(sodium tripolyphosphate), PEG (polyethyleneglicol) and PVP(polyv<strong>in</strong>ylpyrrolidon) as dispersants (Figure 1).Sediment height (mm)130125120115110105100950 0.5 1 1.5 2 2.5 3PVP(wt. %)Time (h)Figure 1. Effect of amount of (a) PVP on sedimentation heightAccord<strong>in</strong>g to the result, the b<strong>in</strong>ary solvent system of ethanoland toluene (at a volume ratio 70:30) with 3 wt.% addition ofPVP shows the most dispersed behavior and is used as amill<strong>in</strong>g medium for mill<strong>in</strong>g studies. The first powder producedby conventional method which has a powder:ball:alcohol ratioof 1:1.5:2 and the other powder produced by high-energymechanical mill<strong>in</strong>g <strong>in</strong> wet medium at 450 rpm for differenttimes up to 40 hours. For the characterization of the particlesize of the milled powders, two different types of method wereused <strong>in</strong> this study. One of these is the dynamic light scatter<strong>in</strong>gmethod, and the average particle size of the milled powders isgiven <strong>in</strong> Fig. 2-3.The second method, BET, was used tomeasure the surface area of the powders an the mean particlesize of (DRBETR). Comparison of the results of both methodsshows that even though the particle size of powders decrease0.51.02.52.01.53.0by <strong>in</strong>creas<strong>in</strong>g mill<strong>in</strong>g time, from sample C to N50. There wasobserved differences <strong>in</strong> results. These differences can beexpla<strong>in</strong>ed by the formation of agglomerates which are difficultto deform, even us<strong>in</strong>g ultrasonic treatment <strong>in</strong> the dynamic lightscatter<strong>in</strong>g method. However, <strong>in</strong> the BET method the diffusionof NR2R gas <strong>in</strong>to agglomerates is possible, giv<strong>in</strong>g more realisticresults than the dynamic light scatter<strong>in</strong>g method.Particle Size (nm)7006005004003002001000Conventional51020Time (h)Figure 2. Particle size analyse of powder by dynamic light scatter<strong>in</strong>gmethodParticle Size (nm)250200150100500Conventional51020Time (h)Figure 3. Particle size analyse of powder by BETAs a consequence, the high energy mill<strong>in</strong>g <strong>in</strong> a wet mediumsystem is an effective way to produce nano- -SiAlONstart<strong>in</strong>g powders. The most efficient mill<strong>in</strong>g medium for the-SiAlON suspensions is <strong>in</strong>vestigated as 70 vol. % Ethanol-30vol. % Toluen with the addition of 3 wt% PVP. The m<strong>in</strong>imum-SiAlON powder size was obta<strong>in</strong>ed as 92 nm after 50 hmill<strong>in</strong>g <strong>in</strong> this medium.This work was supported by TUBITAK under Grant No.108T661.*Correspond<strong>in</strong>g author: skurama@anadolu.edu.tr[1]. Bulic, F., Zalite, I., Zhil<strong>in</strong>ska, N., 2004. Comparison of plasma-chemicalsynthesised SiAlON nano-powder and conventional prepared SiAlONpowder, Journal of the European Ceramic Society, 24: 3303-3306.[2]. Li, Q., Zhang, C., Komeya, K., Tatami, J., Meguro, T., Gao, L., 2003.Nano powders of -SiAlON carbothermally produced via a sol-gel process,Journal of Materials Science Letters, 22: 885-887.[3]. Xu, X., Nishimura, T., Hirosaki, N., Xie, R., Yamamoto, Y., Tanaka, H.,2005. Fabrication of -SiAlON nanoceramics by high-energy mechanicalmill<strong>in</strong>g and spark plasma s<strong>in</strong>ter<strong>in</strong>g, Nanotechnology, 16 : 1569-1573.[4] Eser, O. and Kurama, S., (2010). The effect of the wet-mill<strong>in</strong>g process ons<strong>in</strong>ter<strong>in</strong>g temperature and the amount of additive of SiAlON ceramics,Ceramics International, doi:10.1016/j.ceram<strong>in</strong>t.2009.12.025.[5] Eser, O., Kurama, S. and Gunkaya, G., 2010. The production of -SiAlON ceramics with low amount of additive, at low s<strong>in</strong>ter<strong>in</strong>g temperature,J. The European ceram. Soc,. doi: 10.1016/j.jeurceramsoc.2010.01.024.3030404050506th Nanoscience and Nanotechnology Conference, zmir, 2010 655
- Page 1: Poster Presentations3rd Day17 June
- Page 4 and 5: Determination of Dielectric Anisotr
- Page 7 and 8: Poster Session, Thursday, June 17Th
- Page 9 and 10: PP mPP vs.P =P,PP (1)P andPoster Se
- Page 11 and 12: PP mPP vs.P =P,PP (1)P andPoster Se
- Page 13 and 14: PP andPoster Session, Thursday, Jun
- Page 15 and 16: Poster Session, Thursday, June 17Th
- Page 17 and 18: PP and770 772 774 776 778 780 782 7
- Page 19 and 20: Poster Session, Thursday, June 17Th
- Page 21 and 22: Poster Session, Thursday, June 17Th
- Page 23 and 24: P25,Poster Session, Thursday, June
- Page 25 and 26: PP TOBBPoster Session, Thursday, Ju
- Page 27 and 28: PisPPisisisP,PisPoster Session, Thu
- Page 29 and 30: U NeslihanPPPPoster Session, Thursd
- Page 31 and 32: Poster Session, Thursday, June 17Th
- Page 33 and 34: PPPoster Session, Thursday, June 17
- Page 35 and 36: PPoster Session, Thursday, June 17T
- Page 37 and 38: P onP viaPP wereP upPoster Session,
- Page 39 and 40: P ·cm.PVPPPsPPPPP andPoster Sessio
- Page 41 and 42: Poster Session, Thursday, June 17Th
- Page 43: PPoster Session, Thursday, June 17T
- Page 47 and 48: Poster Session, Thursday, June 17Th
- Page 49 and 50: PErkanPoster Session, Thursday, Jun
- Page 51 and 52: Poster Session, Thursday, June 17Th
- Page 53 and 54: Poster Session, Thursday, June 17Th
- Page 55 and 56: PPPP andPoster Session, Thursday, J
- Page 57 and 58: Poster Session, Thursday, June 17Th
- Page 59 and 60: Poster Session, Thursday, June 17Th
- Page 61 and 62: T PeptideTPP,PP,PP andTT2429TTTTTT
- Page 63 and 64: Poster Session, Thursday, June 17Th
- Page 65 and 66: PPoster Session, Thursday, June 17T
- Page 67 and 68: Poster Session, Thursday, June 17Th
- Page 69 and 70: PPPoster Session, Thursday, June 17
- Page 71 and 72: Poster Session, Thursday, June 17Th
- Page 73 and 74: Poster Session, Thursday, June 17Th
- Page 75 and 76: PT AdditionalT ThePoster Session, T
- Page 77 and 78: Poster Session, Thursday, June 17Th
- Page 79 and 80: Poster Session, Thursday, June 17Th
- Page 81 and 82: Poster Session, Thursday, June 17Th
- Page 83 and 84: PPoster Session, Thursday, June 17T
- Page 85 and 86: Poster Session, Thursday, June 17Th
- Page 87 and 88: PPPoster Session, Thursday, June 17
- Page 89 and 90: Poster Session, Thursday, June 17Hu
- Page 91 and 92: Poster Session, Thursday, June 17Th
- Page 93 and 94: PPPPPPoster Session, Thursday, June
- Page 95 and 96:
Poster Session, Thursday, June 17Th
- Page 97 and 98:
Poster Session, Thursday, June 17Th
- Page 99 and 100:
Poster Session, Thursday, June 17Th
- Page 101 and 102:
PPoster Session, Thursday, June 17T
- Page 103 and 104:
Poster Session, Thursday, June 17Th
- Page 105 and 106:
PPPPPPPoster Session, Thursday, Jun
- Page 107 and 108:
Poster Session, Thursday, June 17Th
- Page 109 and 110:
PPPR2R PIN(80)PPgPP OzlemPPoster Se
- Page 111 and 112:
Poster Session, Thursday, June 17Th
- Page 113 and 114:
Poster Session, Thursday, June 17Th
- Page 115 and 116:
P onPP toP coordinatedPPoster Sessi
- Page 117 and 118:
PPPPP,PP,P(PR RmPoster Session, Thu
- Page 119 and 120:
Poster Session, Thursday, June 17Th
- Page 121 and 122:
Poster Session, Thursday, June 17Th
- Page 123 and 124:
PP InstitutePP DepartmentPoster Ses
- Page 125 and 126:
andPCPPoster Session, Thursday, Jun
- Page 127 and 128:
PP scatteringPYusufPP Corresponding
- Page 129 and 130:
PP toPoster Session, Thursday, June
- Page 131 and 132:
PP andPoster Session, Thursday, Jun
- Page 133 and 134:
PPPPoster Session, Thursday, June 1
- Page 135 and 136:
PPoster Session, Thursday, June 17T
- Page 137 and 138:
PPP andP (.cm).Poster Session, Thur
- Page 139 and 140:
PP tiltP andP editionPoster Session
- Page 141 and 142:
PP andPPoster Session, Thursday, Ju
- Page 143 and 144:
Poster Session, Thursday, June 17Th
- Page 145 and 146:
PP forP forP edit.PPoster Session,
- Page 147 and 148:
Poster Session, Thursday, June 17Th
- Page 149 and 150:
Poster Session, Thursday, June 17Th
- Page 151 and 152:
PP ionicPP ,PPoster Session, Thursd
- Page 153 and 154:
PP lightPoster Session, Thursday, J
- Page 155 and 156:
Poster Session, Thursday, June 17Th
- Page 157 and 158:
PPoster Session, Thursday, June 17T
- Page 159 and 160:
Poster Session, Thursday, June 17Th
- Page 161 and 162:
PandPoster Session, Thursday, June
- Page 163 and 164:
Poster Session, Thursday, June 17 T
- Page 165 and 166:
PPPoster Session, Thursday, June 17
- Page 167 and 168:
PPoster Session, Thursday, June 17T
- Page 169 and 170:
PPoster Session, Thursday, June 17T
- Page 171 and 172:
PPoster Session, Thursday, June 17T
- Page 173 and 174:
PP DepartmentNanoscienceTPPoster Se
- Page 175 and 176:
Poster Session, Thursday, June 17Th
- Page 177 and 178:
Poster Session, Thursday, June 17Th
- Page 179 and 180:
PPPoster Session, Thursday, June 17
- Page 181 and 182:
PPPPPoster Session, Thursday, June
- Page 183 and 184:
PPPPoster Session, Thursday, June 1
- Page 185 and 186:
PPoster Session, Thursday, June 17T
- Page 187 and 188:
PPoster Session, Thursday, June 17T
- Page 189 and 190:
PPoster Session, Thursday, June 17T
- Page 191 and 192:
Poster Session, Thursday, June 17Th
- Page 193 and 194:
Poster Session, Thursday, June 17Th
- Page 195 and 196:
0T0T0T0T AsPPPP werePoster Session,
- Page 197 and 198:
PPoster Session, Thursday, June 17T
- Page 199 and 200:
PPPPPoster Session, Thursday, June
- Page 201 and 202:
PPoster Session, Thursday, June 17T
- Page 203 and 204:
PPoster Session, Thursday, June 17T
- Page 205 and 206:
Poster Session, Thursday, June 17Th
- Page 207 and 208:
PPoster Session, Thursday, June 17T
- Page 209 and 210:
PPoster Session, Thursday, June 17T
- Page 211:
Poster Session, Thursday, June 17AF