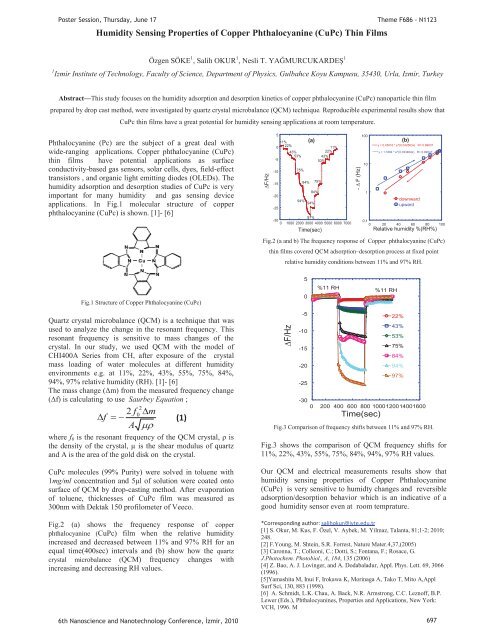
Poster Session, Thursday, June 17Humidity Sens<strong>in</strong>g Properties of Copper Phthalocyan<strong>in</strong>e (CuPc) Th<strong>in</strong> FilmsTheme F686 - N1123Özgen SÖKE 1 , Salih OKUR 1 , Nesli T. YAĞMURCUKARDEŞ 11 Izmir Institute of Technology, Faculty of Science, Department of Physics, Gulbahce Koyu Kampusu, 35430, Urla, Izmir, TurkeyAbstract—This study focuses on the humidity adsorption and desorption k<strong>in</strong>etics of copper phthalocyan<strong>in</strong>e (CuPc) nanoparticle th<strong>in</strong> filmprepared by drop cast method, were <strong>in</strong>vestigated by quartz crystal microbalance (QCM) technique. Reproducible experimental results show thatCuPc th<strong>in</strong> films have a great potential for humidity sens<strong>in</strong>g applications at room temperature.Phthalocyan<strong>in</strong>e (Pc) are the subject of a great deal withwide-rang<strong>in</strong>g applications. Copper phthalocyan<strong>in</strong>e (CuPc)th<strong>in</strong> films have potential applications as surfaceconductivity-based gas sensors, solar cells, dyes, field-effecttransistors , and organic light emitt<strong>in</strong>g diodes (OLEDs). Thehumidity adsorption and desorption studies of CuPc is veryimportant for many humidity and gas sens<strong>in</strong>g deviceapplications. In Fig.1 molecular structure of copperphthalocyan<strong>in</strong>e (CuPc) is shown. [1]- [6]F/Hz50-5-10-15-20-25(a)11%22%11%43%22%53%43%53%75%84% 75%84%94% 94%97%-300 1000 2000 3000 4000 5000 6000 7000Time(sec)- F (Hz)100101y = 0.45016 * e^(0.042563x) R= 0.99637y = 1.1664 * e^(0.033464x) R= 0.99547downwardupward0,10 20 40 60 80 100Relative humidity %(RH%)Fig.2 (a and b) The frequency response of Copper phthalocyan<strong>in</strong>e (CuPc)th<strong>in</strong> films covered QCM adsorption–desorption process at fixed po<strong>in</strong>trelative humidity conditions between 11% and 97% RH.(b)Fig.1 Structure of Copper Phthalocyan<strong>in</strong>e (CuPc)50%11 RH%11 RHQuartz crystal microbalance (QCM) is a technique that wasused to analyze the change <strong>in</strong> the resonant frequency. Thisresonant frequency is sensitive to mass changes of thecrystal. In our study, we used QCM with the model ofCHI400A Series from CH, after exposure of the crystalmass load<strong>in</strong>g of water molecules at different humidityenvironments e.g. at 11%, 22%, 43%, 55%, 75%, 84%,94%, 97% relative humidity (RH). [1]- [6]The mass change (Δm) from the measured frequency change(Δf) is calculat<strong>in</strong>g to use Saurbey Equation ;22 f0mf A (1)where f 0 is the resonant frequency of the QCM crystal, ρ isthe density of the crystal, μ is the shear modulus of quartzand A is the area of the gold disk on the crystal.CuPc molecules (99% Purity) were solved <strong>in</strong> toluene with1mg/ml concentration and 5μl of solution were coated ontosurface of QCM by drop-cast<strong>in</strong>g method. After evaporationof toluene, thicknesses of CuPc film was measured as300nm with Dektak 150 profilometer of Veeco.Fig.2 (a) shows the frequency response of copperphthalocyan<strong>in</strong>e (CuPc) film when the relative humidity<strong>in</strong>creased and decreased between 11% and 97% RH for anequal time(400sec) <strong>in</strong>tervals and (b) show how the quartzcrystal microbalance (QCM) frequency changes with<strong>in</strong>creas<strong>in</strong>g and decreas<strong>in</strong>g RH values.F/Hz-5-10-15-20-2522%43%53%75%84%94%97%-300 200 400 600 800 1000120014001600Time(sec)Fig.3 Comparison of frequency shifts between 11% and 97% RH.Fig.3 shows the comparison of QCM frequency shifts for11%, 22%, 43%, 55%, 75%, 84%, 94%, 97% RH values.Our QCM and electrical measurements results show thathumidity sens<strong>in</strong>g properties of Copper Phthalocyan<strong>in</strong>e(CuPc) is very sensitive to humidty changes and reversibleadsorption/desorption behavior which is an <strong>in</strong>dicative of agood humidity sensor even at room temprature.*Correspond<strong>in</strong>g author: salihokur@iyte.edu.tr[1] S. Okur, M. Kus, F. Özel, V. Aybek, M. Yilmaz, Talanta, 81;1-2; 2010;248.[2] F.Young, M. Shte<strong>in</strong>, S.R. Forrest, Nature Mater.4,37,(2005)[3] Caronna, T.; Colleoni, C.; Dotti, S.; Fontana, F.; Rosace, G.J.Photochem. Photobiol., A, 184, 135 (2006)[4] Z. Bao, A. J. Lov<strong>in</strong>ger, and A. Dodabaladur, Appl. Phys. Lett. 69, 3066(1996).[5]Yamashita M, Inui F, Irokawa K, Mor<strong>in</strong>aga A, Tako T, Mito A,ApplSurf Sci, 130, 883 (1998).[6] A. Schmidt, L.K. Chau, A. Back, N.R. Armstrong, C.C. Leznoff, B.P.Lewer (Eds.), Phthalocyan<strong>in</strong>es, Properties and Applications, New York:VCH, 1996. M6th Nanoscience and Nanotechnology Conference, zmir, 2010 697
Poster Session, Thursday, June 17Humidity Sens<strong>in</strong>g Investigation of ZnO Nanostructures Us<strong>in</strong>g QCM TechniqueNurdan Asar 1 , Nesli Tekguzel Yagmurcukardes 2 , Ayse Erol 1 , Salih Okur 2 , M. Cet<strong>in</strong> Arikan 11 Istanbul University Faculty of Science Physics Department 34134 Vezneciler, Istanbul, Turkey2 Izmir Institute of Technology 35430 Urla, Izmir, TurkeyTheme F686 - N1123Abstract: ZnO nanostructures were synthesized via chemical sol-gel method <strong>in</strong> two different morphologies. Their humidity sens<strong>in</strong>g properties were <strong>in</strong>vestigated byus<strong>in</strong>g Quartz Crystal Microbalance (QCM) technique. It was found that the frequency shift of the ZnO nanostructures coated on QCM <strong>in</strong>creases with <strong>in</strong>creas<strong>in</strong>g relativehumidity between 33-77 % at room temperature. The results show that humidity sens<strong>in</strong>g properties are strongly dependent on morphology of the nanostructures.ZnO is one of the most important promis<strong>in</strong>g metal oxidesemiconductors for gas/vapour/humidity sens<strong>in</strong>g applications and haspronounced sensitivity to gases such as NH 3 , NO 2 , CO, H 2 , ethanoland humidity [1-4]. It has been observed that ZnO nanostructuressynthesized <strong>in</strong> different morphologies compared with its th<strong>in</strong> film orbulk counterparts have much more sensitivity due to their highsurface to volume ratio and more chemically active centers [5].In this study we synthesized ZnO nanostructures by us<strong>in</strong>g chemicalsol-gel method. Crystal structure and morphology of ZnOnanostructures synthesized <strong>in</strong> different experimental conditions werecharacterized by X-Ray Diffraction (XRD) and Scann<strong>in</strong>g ElectronMicroscopy (SEM).(a)(a)Samples S2 and S3 were synthesized with different molarities of Zn +2and OH - solutions. Samples dried <strong>in</strong> ambient air for 24 hours. As seenfrom the Figures 1 (a) and (b), the morphology of S2 is nanoparticlewhile the morphology of S3 is nanowire and both structures havediameter as ~ 20 nm. XRD patterns showed that both samples arecrystallized <strong>in</strong> hexagonal wurtzite structure.Humidity sens<strong>in</strong>g <strong>in</strong>vestigations of ZnO nanostructures werecarried out us<strong>in</strong>g Quartz Crystal Microbalance (QCM) technique.Samples dispersed <strong>in</strong> ethanol were dropped on quartz crystal andexposed to various saturated salt solutions. The frequency responsesof the ZnO nanostructure sensors to relative humidity chang<strong>in</strong>gbetween 33-77% RH were measured at room temperature. Relativehumidity was recorded by commercial sensor simultaneously.(b)Figure 2: Frequency responses of S2 and S3 sensors under 33 – 77% relative humidityexposure at room temperature.(c)(d)Figure 2 shows response and recovery curves of the sensors. When%RH was decreased from 77 to 33%, frequencies of the sensors werebackshifted to their <strong>in</strong>itial values. In comparison with nanowires,nanoparticles showed larger frequency shift.The experimental results demonstrated that ZnO nanoparticles aremore sensitive to humidity changes compared to nanowires due tohav<strong>in</strong>g high surface to volume ratio and much more chemically activecenters.This work was supported by Scientific Research ProjectsCoord<strong>in</strong>ation Unit of Istanbul University. Project number 4907.Figure 1: (a-b) SEM images and (c-d) XRD patterns of samples S2 and S3, respectively.[1] Hongsith N., Choopun S., Mangkorntong P.,Mangkorntong N., 2005. CMU. J.Special issue on nanotechnology. vol. 4 No. 1: 15-20.[2] Sadek A. Z., Choopun S., Wlodarski W., Ippolito S. J., Kalantar Zadeh K., 2007.IEEE Sensors Journal. Vol. 7, No. 6.[3] Krishnakumar T., Jayaprakash R., P<strong>in</strong>na N., Donato N., Bonavita A., Micali G. andNeri G., 2009. Sensors & Actuators: B. 143, 198.[4] Qi Q., Zhang T., Yu Q., Wang R., Zeng Y., Liu L., Yang H., 2008. Sensors andActuators B: Chemical. 638-643.[5] Hongsith N., Viriyaworasakul C., Mangkorntong P. , Mangkorntong N. , ChoopunS., 2008 Ceramics International 34: 823–826.6th Nanoscience and Nanotechnology Conference, zmir, 2010 698
- Page 1:
Poster Presentations3rd Day17 June
- Page 4 and 5:
Determination of Dielectric Anisotr
- Page 7 and 8:
Poster Session, Thursday, June 17Th
- Page 9 and 10:
PP mPP vs.P =P,PP (1)P andPoster Se
- Page 11 and 12:
PP mPP vs.P =P,PP (1)P andPoster Se
- Page 13 and 14:
PP andPoster Session, Thursday, Jun
- Page 15 and 16:
Poster Session, Thursday, June 17Th
- Page 17 and 18:
PP and770 772 774 776 778 780 782 7
- Page 19 and 20:
Poster Session, Thursday, June 17Th
- Page 21 and 22:
Poster Session, Thursday, June 17Th
- Page 23 and 24:
P25,Poster Session, Thursday, June
- Page 25 and 26:
PP TOBBPoster Session, Thursday, Ju
- Page 27 and 28:
PisPPisisisP,PisPoster Session, Thu
- Page 29 and 30:
U NeslihanPPPPoster Session, Thursd
- Page 31 and 32:
Poster Session, Thursday, June 17Th
- Page 33 and 34:
PPPoster Session, Thursday, June 17
- Page 35 and 36:
PPoster Session, Thursday, June 17T
- Page 37 and 38: P onP viaPP wereP upPoster Session,
- Page 39 and 40: P ·cm.PVPPPsPPPPP andPoster Sessio
- Page 41 and 42: Poster Session, Thursday, June 17Th
- Page 43 and 44: PPoster Session, Thursday, June 17T
- Page 45 and 46: PPoster Session, Thursday, June 17T
- Page 47 and 48: Poster Session, Thursday, June 17Th
- Page 49 and 50: PErkanPoster Session, Thursday, Jun
- Page 51 and 52: Poster Session, Thursday, June 17Th
- Page 53 and 54: Poster Session, Thursday, June 17Th
- Page 55 and 56: PPPP andPoster Session, Thursday, J
- Page 57 and 58: Poster Session, Thursday, June 17Th
- Page 59 and 60: Poster Session, Thursday, June 17Th
- Page 61 and 62: T PeptideTPP,PP,PP andTT2429TTTTTT
- Page 63 and 64: Poster Session, Thursday, June 17Th
- Page 65 and 66: PPoster Session, Thursday, June 17T
- Page 67 and 68: Poster Session, Thursday, June 17Th
- Page 69 and 70: PPPoster Session, Thursday, June 17
- Page 71 and 72: Poster Session, Thursday, June 17Th
- Page 73 and 74: Poster Session, Thursday, June 17Th
- Page 75 and 76: PT AdditionalT ThePoster Session, T
- Page 77 and 78: Poster Session, Thursday, June 17Th
- Page 79 and 80: Poster Session, Thursday, June 17Th
- Page 81 and 82: Poster Session, Thursday, June 17Th
- Page 83 and 84: PPoster Session, Thursday, June 17T
- Page 85 and 86: Poster Session, Thursday, June 17Th
- Page 87: PPPoster Session, Thursday, June 17
- Page 91 and 92: Poster Session, Thursday, June 17Th
- Page 93 and 94: PPPPPPoster Session, Thursday, June
- Page 95 and 96: Poster Session, Thursday, June 17Th
- Page 97 and 98: Poster Session, Thursday, June 17Th
- Page 99 and 100: Poster Session, Thursday, June 17Th
- Page 101 and 102: PPoster Session, Thursday, June 17T
- Page 103 and 104: Poster Session, Thursday, June 17Th
- Page 105 and 106: PPPPPPPoster Session, Thursday, Jun
- Page 107 and 108: Poster Session, Thursday, June 17Th
- Page 109 and 110: PPPR2R PIN(80)PPgPP OzlemPPoster Se
- Page 111 and 112: Poster Session, Thursday, June 17Th
- Page 113 and 114: Poster Session, Thursday, June 17Th
- Page 115 and 116: P onPP toP coordinatedPPoster Sessi
- Page 117 and 118: PPPPP,PP,P(PR RmPoster Session, Thu
- Page 119 and 120: Poster Session, Thursday, June 17Th
- Page 121 and 122: Poster Session, Thursday, June 17Th
- Page 123 and 124: PP InstitutePP DepartmentPoster Ses
- Page 125 and 126: andPCPPoster Session, Thursday, Jun
- Page 127 and 128: PP scatteringPYusufPP Corresponding
- Page 129 and 130: PP toPoster Session, Thursday, June
- Page 131 and 132: PP andPoster Session, Thursday, Jun
- Page 133 and 134: PPPPoster Session, Thursday, June 1
- Page 135 and 136: PPoster Session, Thursday, June 17T
- Page 137 and 138: PPP andP (.cm).Poster Session, Thur
- Page 139 and 140:
PP tiltP andP editionPoster Session
- Page 141 and 142:
PP andPPoster Session, Thursday, Ju
- Page 143 and 144:
Poster Session, Thursday, June 17Th
- Page 145 and 146:
PP forP forP edit.PPoster Session,
- Page 147 and 148:
Poster Session, Thursday, June 17Th
- Page 149 and 150:
Poster Session, Thursday, June 17Th
- Page 151 and 152:
PP ionicPP ,PPoster Session, Thursd
- Page 153 and 154:
PP lightPoster Session, Thursday, J
- Page 155 and 156:
Poster Session, Thursday, June 17Th
- Page 157 and 158:
PPoster Session, Thursday, June 17T
- Page 159 and 160:
Poster Session, Thursday, June 17Th
- Page 161 and 162:
PandPoster Session, Thursday, June
- Page 163 and 164:
Poster Session, Thursday, June 17 T
- Page 165 and 166:
PPPoster Session, Thursday, June 17
- Page 167 and 168:
PPoster Session, Thursday, June 17T
- Page 169 and 170:
PPoster Session, Thursday, June 17T
- Page 171 and 172:
PPoster Session, Thursday, June 17T
- Page 173 and 174:
PP DepartmentNanoscienceTPPoster Se
- Page 175 and 176:
Poster Session, Thursday, June 17Th
- Page 177 and 178:
Poster Session, Thursday, June 17Th
- Page 179 and 180:
PPPoster Session, Thursday, June 17
- Page 181 and 182:
PPPPPoster Session, Thursday, June
- Page 183 and 184:
PPPPoster Session, Thursday, June 1
- Page 185 and 186:
PPoster Session, Thursday, June 17T
- Page 187 and 188:
PPoster Session, Thursday, June 17T
- Page 189 and 190:
PPoster Session, Thursday, June 17T
- Page 191 and 192:
Poster Session, Thursday, June 17Th
- Page 193 and 194:
Poster Session, Thursday, June 17Th
- Page 195 and 196:
0T0T0T0T AsPPPP werePoster Session,
- Page 197 and 198:
PPoster Session, Thursday, June 17T
- Page 199 and 200:
PPPPPoster Session, Thursday, June
- Page 201 and 202:
PPoster Session, Thursday, June 17T
- Page 203 and 204:
PPoster Session, Thursday, June 17T
- Page 205 and 206:
Poster Session, Thursday, June 17Th
- Page 207 and 208:
PPoster Session, Thursday, June 17T
- Page 209 and 210:
PPoster Session, Thursday, June 17T
- Page 211:
Poster Session, Thursday, June 17AF