198 Topics in Current Chemistry Editorial Board: A. de Meijere KN ...
198 Topics in Current Chemistry Editorial Board: A. de Meijere KN ...
198 Topics in Current Chemistry Editorial Board: A. de Meijere KN ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
122 R.E. Melén<strong>de</strong>z · A.D. Hamilton<br />
3.4<br />
Weak Interactions<br />
In addition to strong hydrogen bonds, the use of weak <strong>in</strong>termolecular <strong>in</strong>teractions<br />
has been predicted to yield new and excit<strong>in</strong>g results <strong>in</strong> the future of crystal<br />
eng<strong>in</strong>eer<strong>in</strong>g. Hydrogen bonds of type C-H◊◊◊O(N) and O(N)-H◊◊◊p as well as<br />
polarization <strong>in</strong>duced iodo◊◊◊nitro <strong>in</strong>teractions have been exam<strong>in</strong>ed and used <strong>in</strong><br />
<strong>de</strong>sign<strong>in</strong>g new solids [68].<br />
The study of C-H◊◊◊O hydrogen bonds has become <strong>in</strong>creas<strong>in</strong>gly important <strong>in</strong><br />
our un<strong>de</strong>rstand<strong>in</strong>g of the pack<strong>in</strong>g of molecules <strong>in</strong> crystals [69]. In the past,<br />
observation of higher boil<strong>in</strong>g [70] and melt<strong>in</strong>g [71] po<strong>in</strong>ts <strong>in</strong> organic compounds<br />
have been ascribed to weak C-H◊◊◊O hydrogen bond formation. In a<br />
survey of 113 published neutron diffraction organic crystal structures, Taylor<br />
and Kennard [72] conclu<strong>de</strong>d that C-H◊◊◊O, C-H◊◊◊N, and C-H◊◊◊Cl <strong>in</strong>teractions<br />
are more likely to be attractive than repulsive. In their study, typical C◊◊◊O(N)<br />
distances occur <strong>in</strong> the range of 3–4 Å and the hydrogen bond angles are observed<br />
between 100–180°, but with a major cluster around 150–160° (see also<br />
the articles by J.P. Glusker and A.Nangia and G.R. Desiraju <strong>in</strong> this volume).<br />
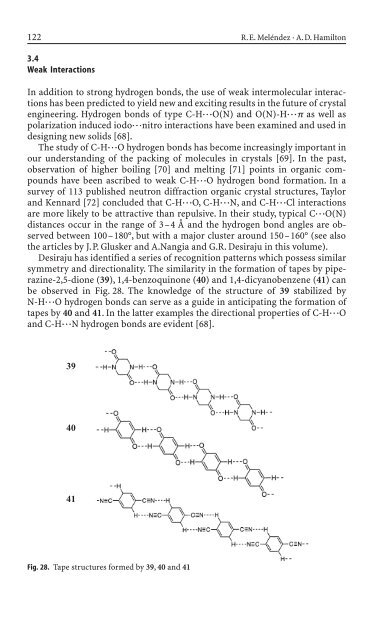
Desiraju has i<strong>de</strong>ntified a series of recognition patterns which possess similar<br />
symmetry and directionality. The similarity <strong>in</strong> the formation of tapes by piperaz<strong>in</strong>e-2,5-dione<br />
(39), 1,4-benzoqu<strong>in</strong>one (40) and 1,4-dicyanobenzene (41) can<br />
be observed <strong>in</strong> Fig. 28. The knowledge of the structure of 39 stabilized by<br />
N-H◊◊◊O hydrogen bonds can serve as a gui<strong>de</strong> <strong>in</strong> anticipat<strong>in</strong>g the formation of<br />
tapes by 40 and 41. In the latter examples the directional properties of C-H◊◊◊O<br />
and C-H◊◊◊N hydrogen bonds are evi<strong>de</strong>nt [68].<br />
39<br />
40<br />
41<br />
Fig. 28. Tape structures formed by 39, 40 and 41