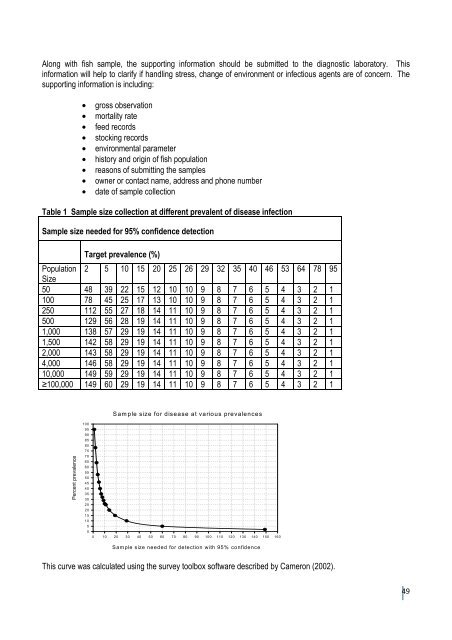
Along with fish sample, the support<strong>in</strong>g <strong>in</strong>formation should be submitted to the diagnostic laboratory. This<strong>in</strong>formation will help to clarify if handl<strong>in</strong>g stress, change <strong>of</strong> environment or <strong>in</strong>fectious agents are <strong>of</strong> concern. Thesupport<strong>in</strong>g <strong>in</strong>formation is <strong>in</strong>clud<strong>in</strong>g:• gross observation• mortality rate• feed records• stock<strong>in</strong>g records• environmental parameter• history and orig<strong>in</strong> <strong>of</strong> fish population• reasons <strong>of</strong> submitt<strong>in</strong>g the samples• owner or contact name, address and phone number• date <strong>of</strong> sample collectionTable 1 Sample size collection at different prevalent <strong>of</strong> disease <strong>in</strong>fectionSample size needed for 95% confidence detectionTarget prevalence (%)Population 2 5 10 15 20 25 26 29 32 35 40 46 53 64 78 95Size50 48 39 22 15 12 10 10 9 8 7 6 5 4 3 2 1100 78 45 25 17 13 10 10 9 8 7 6 5 4 3 2 1250 112 55 27 18 14 11 10 9 8 7 6 5 4 3 2 1500 129 56 28 19 14 11 10 9 8 7 6 5 4 3 2 11,000 138 57 29 19 14 11 10 9 8 7 6 5 4 3 2 11,500 142 58 29 19 14 11 10 9 8 7 6 5 4 3 2 12,000 143 58 29 19 14 11 10 9 8 7 6 5 4 3 2 14,000 146 58 29 19 14 11 10 9 8 7 6 5 4 3 2 110,000 149 59 29 19 14 11 10 9 8 7 6 5 4 3 2 1≥100,000 149 60 29 19 14 11 10 9 8 7 6 5 4 3 2 1Sample size for disease at various prevalencesPercent prevalence100959085807570656055504540353025201510500 10 20 30 40 50 60 70 80 90 100 110 120 130 140 150 160Sample size needed for detection with 95% confidenceThis curve was calculated us<strong>in</strong>g the survey toolbox s<strong>of</strong>tware described by Cameron (2002).49
Live sample collection for transportationFish should be packed <strong>in</strong> secured conta<strong>in</strong>ers such as double plastic bags or polystyrene box. Polystyrene box issuitable to pack the sp<strong>in</strong>y fish species which requires a durable conta<strong>in</strong>er and when the dest<strong>in</strong>ation is not far.Double plastic bags are more popular for long distance transportation <strong>in</strong>clud<strong>in</strong>g by airways. Water can be filled toone third <strong>of</strong> the conta<strong>in</strong>er capacity with the rema<strong>in</strong><strong>in</strong>g 2/3 volume <strong>in</strong>flated with oxygen. The bags should be tightlysealed with rubber bands and packed <strong>in</strong>side a polystyrene box or cardboard box l<strong>in</strong>ed with styr<strong>of</strong>oam. Ice packsmay be required for a long transportation. The volume <strong>of</strong> water to fish volume is particularly important for live fishbe<strong>in</strong>g shipped for ectoparasite exam<strong>in</strong>ation, so advance check<strong>in</strong>g with the diagnostic laboratory is recommended.To shorten the time between removal <strong>of</strong> fish from water and transportation, samples should be collected as closeas to the shipp<strong>in</strong>g time and early <strong>in</strong>form the diagnostic laboratory.Tissue sample collectionPrior to fixation, fish should be humanly killed. Small fish can be done by decapitation, while the larger fish isdone with a overdose <strong>of</strong> anaesthetics. Very small fish can be wholly immersed <strong>in</strong>to the fixative agent <strong>in</strong> a m<strong>in</strong>imum<strong>of</strong> 10:1 (fixative: fish) volume ratio. For large fish, the body cavity should be split open and displaced the visceraorgans to allow maximum penetration <strong>of</strong> the fixative. The organs can be possibly removed and fixed separatelywith the 10:1 (fixative: sample) volume ratio. Most tissues require a m<strong>in</strong>imum <strong>of</strong> 24-48 h fixation time. Howeverfor some specific diagnostic techniques, the length <strong>of</strong> fixation may be varied. For <strong>in</strong>stance, the samples collectedfor DNA-based technique analysis, the fixation time should not exceed 24 h as it may cause difficulty to recoverthe DNA from sample. So, it is highly recommended to get some advice for the diagnostic laboratory before theprocedure.The most suitable fixative for preservation <strong>of</strong> f<strong>in</strong>fish for histopathological study is phosphate buffered formal<strong>in</strong>which can be prepared as follow<strong>in</strong>g:37-40% Formaldehyde 100mlTap water 900 mlNaH 2 PO 4 .H 2 O 4.0 gNa 2 HPO 4 6.5 gGeneral diagnosisExternal exam<strong>in</strong>ationPlace the specimens on the clean flat board and keep them moist dur<strong>in</strong>g exam<strong>in</strong>ation. Exam<strong>in</strong>e external bodysurface, body form, abdom<strong>in</strong>al form, sk<strong>in</strong> coloration, eye appearance, and record any abnormality. The entiresurface should be exam<strong>in</strong>ed <strong>in</strong>clud<strong>in</strong>g opercular and oral cavities, gills and f<strong>in</strong>s.Scrape along the body trunk <strong>of</strong> specimen with coverslip and place on a slide with a drop <strong>of</strong> water. Exam<strong>in</strong>e thesample under a light microscope. Gill samples are also taken and exam<strong>in</strong>e <strong>in</strong> the same method.Internal exam<strong>in</strong>ationWithdraw blood samples from a caudal peduncle, where the fish is too small to do so, the caudal peduncle can bechopped <strong>of</strong>f to collect the blood sample. Place a small drop <strong>of</strong> blood and smear on the slide and observe underthe light microscope for any parasite <strong>in</strong>fection.Open up the abdom<strong>in</strong>al cavity. Observe any lesion and abnormality <strong>of</strong> the visceral organs such as discoloration,enlargement, ascites, oedema, lesion and nodules. The organ can be aseptically collected for bacteriogical andvirological exam<strong>in</strong>ation.50
- Page 1 and 2: Training of TrainersProgramme3-7 Au
- Page 3 and 4: Table of Contents1. Preface 42. Sen
- Page 5 and 6: knowledge about the activities carr
- Page 7 and 8: iosphere, in that it is essentially
- Page 10 and 11: Importance in narrowing the supply
- Page 12 and 13: Figure 7: The trend in aquaculture
- Page 14: aquaculture has been a success thus
- Page 17 and 18: pathogen transfer is generally cons
- Page 19 and 20: • It highlights the importance a
- Page 21 and 22: particular animal are identified, t
- Page 23: Knowledge at the bottom of the pyra
- Page 26 and 27: farmer innovation process. In this
- Page 28 and 29: • Rapport building• Working str
- Page 30 and 31: shrimp farming is undertaken in the
- Page 32 and 33: Reintjes, C and Hiemstra, W. 1989 F
- Page 34 and 35: • An increase in reliable product
- Page 36 and 37: Principles of AACC are to: 1) Facil
- Page 38 and 39: Maintaining genetic quality of fish
- Page 40 and 41: Where F is the percent increase in
- Page 42 and 43: o G=(0.95) 1/10G =0.994883803Figure
- Page 44 and 45: Aquaculture extension and training
- Page 46 and 47: ResearchExtensionFarmersFlow of inf
- Page 48 and 49: view of outside world than before a
- Page 52 and 53: Fungal examinationFungal infection
- Page 54 and 55: few different pathogen strains. The
- Page 56 and 57: Major finfish diseases in Asia and
- Page 58 and 59: A great number and diversity of ani
- Page 60 and 61: • -oxidase test positive• -resi
- Page 62 and 63: ReferencesAustin, B. and D. Austin.
- Page 64 and 65: Accessing better markets-improving
- Page 66 and 67: Owing to its importance in generati
- Page 68 and 69: adopt standards for responsible shr
- Page 70: Tokrisna R, Benheam W., 1995. Gain
- Page 73 and 74: of the temperature rise that were d
- Page 75 and 76: Principles of developing, validatin
- Page 77 and 78: such as determinants, exposures and
- Page 79 and 80: How to promote adoption of BMPs?Pro
- Page 81 and 82: economic sustainability. BMPs need
- Page 83 and 84: Once you understand your stakeholde
- Page 85 and 86: As conventional approaches such as
- Page 87 and 88: Printed publicationsPrinted publica
- Page 89 and 90: Set up a blog or website for your w
- Page 91 and 92: If you have a website, all you need
- Page 93 and 94: ought about increased profits among
- Page 95 and 96: • It is a farming system the prod
- Page 97 and 98: Annex 1. The area surveyed for obta
- Page 99 and 100: contribute substantially to maintai
- Page 101 and 102:
seawater, but in fresh water its le
- Page 103 and 104:
type of ingredients used, since som
- Page 105 and 106:
This is a new approach used to buil
- Page 107 and 108:
New, M.B., A. G. J. Tacon and I. Cs
- Page 109 and 110:
later stages it has become a much m
- Page 111 and 112:
c. Improved information exchange an
- Page 113 and 114:
implemented by both societies. Inte
- Page 115 and 116:
With better informed farmers, the s
- Page 117 and 118:
According to FAO, it is estimated t
- Page 119 and 120:
Farmer organization as models for p
- Page 121 and 122:
NaCSA disseminates BMPs mainly thro
- Page 123 and 124:
1. Drain the pond water completely
- Page 125 and 126:
Seed transportation and Stocking:
- Page 127 and 128:
7. Presently 100% of the society po
- Page 129 and 130:
would help in sustaining shrimp sec
- Page 131 and 132:
Stress brought about by the capture
- Page 133 and 134:
Enhance women participation in aqua
- Page 135 and 136:
women in aquaculture and identify a
- Page 137 and 138:
major percentage of staff, the need
- Page 139 and 140:
having more self confidence through
- Page 141 and 142:
Overall status of men and women in
- Page 143 and 144:
Compliance to international standar
- Page 145 and 146:
(ii) to protect human or animal lif
- Page 147 and 148:
c) FAO/WHO Codex Alimentarius Commi
- Page 149 and 150:
Annex 1: List of participantsCountr
- Page 151 and 152:
Annex 2: AgendaDate Time Presentati
- Page 153:
Annex 3: List of resource persons1.