Monograph on the Potential Human Reproductive and ... - OEHHA
Monograph on the Potential Human Reproductive and ... - OEHHA
Monograph on the Potential Human Reproductive and ... - OEHHA
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
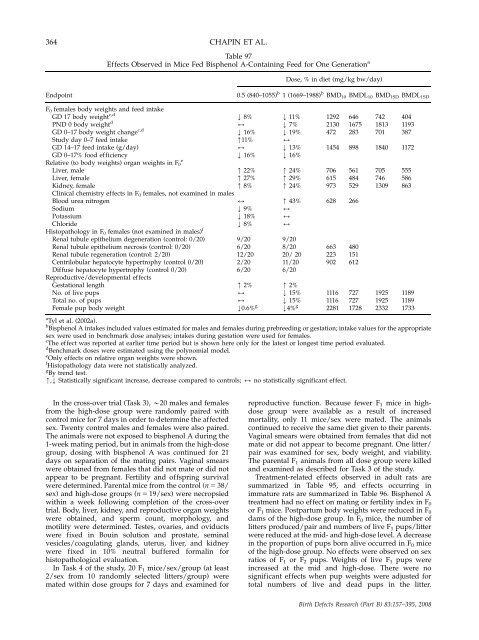
364 CHAPIN ET AL.<br />
Table 97<br />
Effects Observed in Mice Fed Bisphenol A-C<strong>on</strong>taining Feed for One Generati<strong>on</strong> a<br />
Dose, % in diet (mg/kg bw/day)<br />
Endpoint 0.5 (840–1055) b<br />
1 (1669–1988) b BMD10 BMDL10 BMD1SD BMDL1SD<br />
F 0 females body weights <strong>and</strong> feed intake<br />
GD 17 body weight c,d<br />
PND 0 body weight d<br />
GD 0–17 body weight change c,d<br />
Study day 0–7 feed intake<br />
GD 14–17 feed intake (g/day)<br />
GD 0–17% food efficiency<br />
Relative (to body weights) organ weights in F0<br />
Liver, male<br />
Liver, female<br />
Kidney, female<br />
Clinical chemistry effects in F0 females, not examined in males<br />
Blood urea nitrogen<br />
Sodium<br />
Potassium<br />
Chloride<br />
Histopathology in F 0 females (not examined in males) f<br />
Renal tubule epi<strong>the</strong>lium degenerati<strong>on</strong> (c<strong>on</strong>trol: 0/20)<br />
Renal tubule epi<strong>the</strong>lium necrosis (c<strong>on</strong>trol: 0/20)<br />
Renal tubule regenerati<strong>on</strong> (c<strong>on</strong>trol: 2/20)<br />
Centrilobular hepatocyte hypertrophy (c<strong>on</strong>trol 0/20)<br />
Diffuse hepatocyte hypertrophy (c<strong>on</strong>trol 0/20)<br />
<strong>Reproductive</strong>/developmental effects<br />
Gestati<strong>on</strong>al length<br />
No. of live pups<br />
Total no. of pups<br />
Female pup body weight<br />
e<br />
k 8%<br />
2<br />
k 16%<br />
m11%<br />
2<br />
k 16%<br />
m 22%<br />
m 27%<br />
m 8%<br />
2<br />
k 9%<br />
k 18%<br />
k 8%<br />
9/20<br />
6/20<br />
12/20<br />
2/20<br />
6/20<br />
m 2%<br />
2<br />
2<br />
k0.6% g<br />
k 11%<br />
k 7%<br />
k 19%<br />
2<br />
k 13%<br />
k 16%<br />
m 24%<br />
m 29%<br />
m 24%<br />
m 43%<br />
2<br />
2<br />
2<br />
9/20<br />
8/20<br />
20/ 20<br />
11/20<br />
6/20<br />
m 2%<br />
k 15%<br />
k 15%<br />
k4% g<br />
a<br />
Tyl et al. (2002a).<br />
b<br />
Bisphenol A intakes included values estimated for males <strong>and</strong> females during prebreeding or gestati<strong>on</strong>; intake values for <strong>the</strong> appropriate<br />
sex were used in benchmark dose analyses; intakes during gestati<strong>on</strong> were used for females.<br />
c<br />
The effect was reported at earlier time period but is shown here <strong>on</strong>ly for <strong>the</strong> latest or l<strong>on</strong>gest time period evaluated.<br />
d<br />
Benchmark doses were estimated using <strong>the</strong> polynomial model.<br />
e<br />
Only effects <strong>on</strong> relative organ weights were shown.<br />
f<br />
Histopathology data were not statistically analyzed.<br />
g<br />
By trend test.<br />
m,k Statistically significant increase, decrease compared to c<strong>on</strong>trols; 2 no statistically significant effect.<br />
In <strong>the</strong> cross-over trial (Task 3), B20 males <strong>and</strong> females<br />
from <strong>the</strong> high-dose group were r<strong>and</strong>omly paired with<br />
c<strong>on</strong>trol mice for 7 days in order to determine <strong>the</strong> affected<br />
sex. Twenty c<strong>on</strong>trol males <strong>and</strong> females were also paired.<br />
The animals were not exposed to bisphenol A during <strong>the</strong><br />
1-week mating period, but in animals from <strong>the</strong> high-dose<br />
group, dosing with bisphenol A was c<strong>on</strong>tinued for 21<br />
days <strong>on</strong> separati<strong>on</strong> of <strong>the</strong> mating pairs. Vaginal smears<br />
were obtained from females that did not mate or did not<br />
appear to be pregnant. Fertility <strong>and</strong> offspring survival<br />
were determined. Parental mice from <strong>the</strong> c<strong>on</strong>trol (n 5 38/<br />
sex) <strong>and</strong> high-dose groups (n 5 19/sex) were necropsied<br />
within a week following completi<strong>on</strong> of <strong>the</strong> cross-over<br />
trial. Body, liver, kidney, <strong>and</strong> reproductive organ weights<br />
were obtained, <strong>and</strong> sperm count, morphology, <strong>and</strong><br />
motility were determined. Testes, ovaries, <strong>and</strong> oviducts<br />
were fixed in Bouin soluti<strong>on</strong> <strong>and</strong> prostate, seminal<br />
vesicles/coagulating gl<strong>and</strong>s, uterus, liver, <strong>and</strong> kidney<br />
were fixed in 10% neutral buffered formalin for<br />
histopathological evaluati<strong>on</strong>.<br />
In Task 4 of <strong>the</strong> study, 20 F 1 mice/sex/group (at least<br />
2/sex from 10 r<strong>and</strong>omly selected litters/group) were<br />
mated within dose groups for 7 days <strong>and</strong> examined for<br />
1292<br />
2130<br />
472<br />
1454<br />
706<br />
615<br />
973<br />
628<br />
663<br />
223<br />
902<br />
1116<br />
1116<br />
2281<br />
646<br />
1675<br />
283<br />
898<br />
561<br />
484<br />
529<br />
266<br />
480<br />
151<br />
612<br />
727<br />
727<br />
1728<br />
742<br />
1813<br />
701<br />
1840<br />
705<br />
746<br />
1309<br />
1925<br />
1925<br />
2332<br />
404<br />
1193<br />
387<br />
1172<br />
555<br />
586<br />
863<br />
1189<br />
1189<br />
1733<br />
reproductive functi<strong>on</strong>. Because fewer F 1 mice in highdose<br />
group were available as a result of increased<br />
mortality, <strong>on</strong>ly 11 mice/sex were mated. The animals<br />
c<strong>on</strong>tinued to receive <strong>the</strong> same diet given to <strong>the</strong>ir parents.<br />
Vaginal smears were obtained from females that did not<br />
mate or did not appear to become pregnant. One litter/<br />
pair was examined for sex, body weight, <strong>and</strong> viability.<br />
The parental F 1 animals from all dose group were killed<br />
<strong>and</strong> examined as described for Task 3 of <strong>the</strong> study.<br />
Treatment-related effects observed in adult rats are<br />
summarized in Table 95, <strong>and</strong> effects occurring in<br />
immature rats are summarized in Table 96. Bisphenol A<br />
treatment had no effect <strong>on</strong> mating or fertility index in F0<br />
or F1 mice. Postpartum body weights were reduced in F0<br />
dams of <strong>the</strong> high-dose group. In F 0 mice, <strong>the</strong> number of<br />
litters produced/pair <strong>and</strong> numbers of live F 1 pups/litter<br />
were reduced at <strong>the</strong> mid- <strong>and</strong> high-dose level. A decrease<br />
in <strong>the</strong> proporti<strong>on</strong> of pups born alive occurred in F0 mice<br />
of <strong>the</strong> high-dose group. No effects were observed <strong>on</strong> sex<br />
ratios of F1 or F2 pups. Weights of live F1 pups were<br />
increased at <strong>the</strong> mid <strong>and</strong> high-dose. There were no<br />
significant effects when pup weights were adjusted for<br />
total numbers of live <strong>and</strong> dead pups in <strong>the</strong> litter.<br />
Birth Defects Research (Part B) 83:157–395, 2008