Dealing with salinity in Wheatbelt Valleys - Department of Water
Dealing with salinity in Wheatbelt Valleys - Department of Water
Dealing with salinity in Wheatbelt Valleys - Department of Water
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
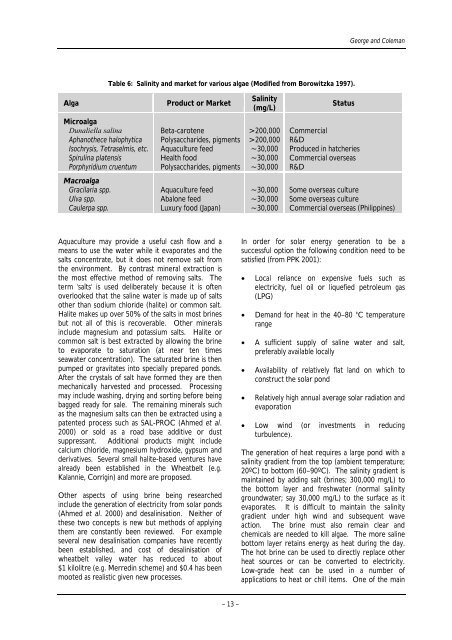
Table 6: Sal<strong>in</strong>ity and market for various algae (Modified from Borowitzka 1997).<br />
Alga Product or Market<br />
Sal<strong>in</strong>ity<br />
(mg/L)<br />
Status<br />
Microalga<br />
Dunaliella sal<strong>in</strong>a Beta-carotene >200,000 Commercial<br />
Aphanothece halophytica Polysaccharides, pigments >200,000 R&D<br />
Isochrysis, Tetraselmis, etc. Aquaculture feed ~30,000 Produced <strong>in</strong> hatcheries<br />
Spirul<strong>in</strong>a platensis Health food ~30,000 Commercial overseas<br />
Porphyridium cruentum Polysaccharides, pigments ~30,000 R&D<br />
George and Coleman<br />
Macroalga<br />
Gracilaria spp. Aquaculture feed ~30,000 Some overseas culture<br />
Ulva spp. Abalone feed ~30,000 Some overseas culture<br />
Caulerpa spp. Luxury food (Japan) ~30,000 Commercial overseas (Philipp<strong>in</strong>es)<br />
Aquaculture may provide a useful cash flow and a<br />
means to use the water while it evaporates and the<br />
salts concentrate, but it does not remove salt from<br />
the environment. By contrast m<strong>in</strong>eral extraction is<br />
the most effective method <strong>of</strong> remov<strong>in</strong>g salts. The<br />
term 'salts' is used deliberately because it is <strong>of</strong>ten<br />
overlooked that the sal<strong>in</strong>e water is made up <strong>of</strong> salts<br />
other than sodium chloride (halite) or common salt.<br />
Halite makes up over 50% <strong>of</strong> the salts <strong>in</strong> most br<strong>in</strong>es<br />
but not all <strong>of</strong> this is recoverable. Other m<strong>in</strong>erals<br />
<strong>in</strong>clude magnesium and potassium salts. Halite or<br />
common salt is best extracted by allow<strong>in</strong>g the br<strong>in</strong>e<br />
to evaporate to saturation (at near ten times<br />
seawater concentration). The saturated br<strong>in</strong>e is then<br />
pumped or gravitates <strong>in</strong>to specially prepared ponds.<br />
After the crystals <strong>of</strong> salt have formed they are then<br />
mechanically harvested and processed. Process<strong>in</strong>g<br />
may <strong>in</strong>clude wash<strong>in</strong>g, dry<strong>in</strong>g and sort<strong>in</strong>g before be<strong>in</strong>g<br />
bagged ready for sale. The rema<strong>in</strong><strong>in</strong>g m<strong>in</strong>erals such<br />
as the magnesium salts can then be extracted us<strong>in</strong>g a<br />
patented process such as SAL-PROC (Ahmed et al.<br />
2000) or sold as a road base additive or dust<br />
suppressant. Additional products might <strong>in</strong>clude<br />
calcium chloride, magnesium hydroxide, gypsum and<br />
derivatives. Several small halite-based ventures have<br />
already been established <strong>in</strong> the <strong>Wheatbelt</strong> (e.g.<br />
Kalannie, Corrig<strong>in</strong>) and more are proposed.<br />
Other aspects <strong>of</strong> us<strong>in</strong>g br<strong>in</strong>e be<strong>in</strong>g researched<br />
<strong>in</strong>clude the generation <strong>of</strong> electricity from solar ponds<br />
(Ahmed et al. 2000) and desal<strong>in</strong>isation. Neither <strong>of</strong><br />
these two concepts is new but methods <strong>of</strong> apply<strong>in</strong>g<br />
them are constantly been reviewed. For example<br />
several new desal<strong>in</strong>isation companies have recently<br />
been established, and cost <strong>of</strong> desal<strong>in</strong>isation <strong>of</strong><br />
wheatbelt valley water has reduced to about<br />
$1 kilolitre (e.g. Merred<strong>in</strong> scheme) and $0.4 has been<br />
mooted as realistic given new processes.<br />
– 13 –<br />
In order for solar energy generation to be a<br />
successful option the follow<strong>in</strong>g condition need to be<br />
satisfied (from PPK 2001):<br />
• Local reliance on expensive fuels such as<br />
electricity, fuel oil or liquefied petroleum gas<br />
(LPG)<br />
• Demand for heat <strong>in</strong> the 40–80 °C temperature<br />
range<br />
• A sufficient supply <strong>of</strong> sal<strong>in</strong>e water and salt,<br />
preferably available locally<br />
• Availability <strong>of</strong> relatively flat land on which to<br />
construct the solar pond<br />
• Relatively high annual average solar radiation and<br />
evaporation<br />
• Low w<strong>in</strong>d (or <strong>in</strong>vestments <strong>in</strong> reduc<strong>in</strong>g<br />
turbulence).<br />
The generation <strong>of</strong> heat requires a large pond <strong>with</strong> a<br />
<strong>sal<strong>in</strong>ity</strong> gradient from the top (ambient temperature;<br />
20ºC) to bottom (60–90ºC). The <strong>sal<strong>in</strong>ity</strong> gradient is<br />
ma<strong>in</strong>ta<strong>in</strong>ed by add<strong>in</strong>g salt (br<strong>in</strong>es; 300,000 mg/L) to<br />
the bottom layer and freshwater (normal <strong>sal<strong>in</strong>ity</strong><br />
groundwater; say 30,000 mg/L) to the surface as it<br />
evaporates. It is difficult to ma<strong>in</strong>ta<strong>in</strong> the <strong>sal<strong>in</strong>ity</strong><br />
gradient under high w<strong>in</strong>d and subsequent wave<br />
action. The br<strong>in</strong>e must also rema<strong>in</strong> clear and<br />
chemicals are needed to kill algae. The more sal<strong>in</strong>e<br />
bottom layer reta<strong>in</strong>s energy as heat dur<strong>in</strong>g the day.<br />
The hot br<strong>in</strong>e can be used to directly replace other<br />
heat sources or can be converted to electricity.<br />
Low-grade heat can be used <strong>in</strong> a number <strong>of</strong><br />
applications to heat or chill items. One <strong>of</strong> the ma<strong>in</strong>