Studies on the use of nano zinc oxide and modified silica in NR, CR ...
Studies on the use of nano zinc oxide and modified silica in NR, CR ...
Studies on the use of nano zinc oxide and modified silica in NR, CR ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Torque (dNm)<br />
9.0<br />
8.5<br />
8.0<br />
7.5<br />
7.0<br />
6.5<br />
6.0<br />
5.5<br />
5.0<br />
4.5<br />
4.0<br />
3.5<br />
3.0<br />
2.5<br />
2.0<br />
1.5<br />
1.0<br />
0.5<br />
0.0<br />
0 5 10 15 20 25 30<br />
Time (m<strong>in</strong>)<br />
Use <strong>of</strong> <strong>nano</strong> <strong>z<strong>in</strong>c</strong> <strong>oxide</strong> <strong>in</strong> styrene butadiene rubber<br />
SBR-ZnO(c) 5phr gum<br />
SBR ZnO(p) 2phr gum<br />
SBR ZnO(s) 2phr gum<br />
SBR ZnO(c) 5phr filled<br />
SBR ZnO(p) 2phr filled<br />
SBR ZnO(s) 2phr filled<br />
SBR ZnO(p) 1phr filled<br />
SBR ZnO(s) 1phr filled<br />
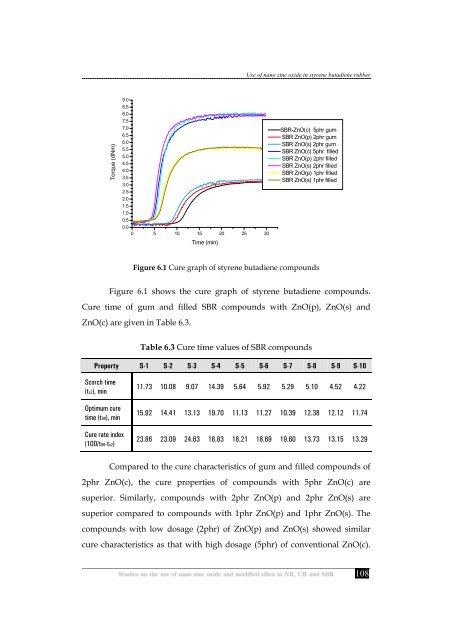
Figure 6.1 Cure graph <strong>of</strong> styrene butadiene compounds<br />
Figure 6.1 shows <strong>the</strong> cure graph <strong>of</strong> styrene butadiene compounds.<br />
Cure time <strong>of</strong> gum <strong>and</strong> filled SBR compounds with ZnO(p), ZnO(s) <strong>and</strong><br />
ZnO(c) are given <strong>in</strong> Table 6.3.<br />
Table 6.3 Cure time values <strong>of</strong> SBR compounds<br />
Property S-1 S-2 S-3 S-4 S-5 S-6 S-7 S-8 S-9 S-10<br />
Scorch time<br />
(ts2), m<strong>in</strong><br />
Optimum cure<br />
time (t90), m<strong>in</strong><br />
Cure rate <strong>in</strong>dex<br />
(100/t90-ts2)<br />
11.73 10.08 9.07 14.39 5.64 5.92 5.29 5.10 4.52 4.22<br />
15.92 14.41 13.13 19.70 11.13 11.27 10.39 12.38 12.12 11.74<br />
23.86 23.09 24.63 18.83 18.21 18.69 19.60 13.73 13.15 13.29<br />
Compared to <strong>the</strong> cure characteristics <strong>of</strong> gum <strong>and</strong> filled compounds <strong>of</strong><br />
2phr ZnO(c), <strong>the</strong> cure properties <strong>of</strong> compounds with 5phr ZnO(c) are<br />
superior. Similarly, compounds with 2phr ZnO(p) <strong>and</strong> 2phr ZnO(s) are<br />
superior compared to compounds with 1phr ZnO(p) <strong>and</strong> 1phr ZnO(s). The<br />
compounds with low dosage (2phr) <strong>of</strong> ZnO(p) <strong>and</strong> ZnO(s) showed similar<br />
cure characteristics as that with high dosage (5phr) <strong>of</strong> c<strong>on</strong>venti<strong>on</strong>al ZnO(c).<br />
108