Principles of Plant Genetics and Breeding
Principles of Plant Genetics and Breeding
Principles of Plant Genetics and Breeding
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
268 CHAPTER 15<br />
that the agencies typically evaluate for every product that falls under their purview. All <strong>of</strong> the agencies evaluate a detailed<br />
molecular <strong>and</strong> genetic characterization <strong>of</strong> the product to obtain information about the identity <strong>of</strong> the GE plant <strong>and</strong> confirm<br />
that the inserted gene is functioning as intended. In addition, the FDA <strong>and</strong> USDA-APHIS examine plant composition to gauge<br />
unintended, pleiotropic changes due to transgenesis, <strong>and</strong> the FDA <strong>and</strong> EPA evaluate the allergenicity potential <strong>of</strong> the expressed<br />
protein(s).<br />
USDA-APHIS determines the potential <strong>of</strong> a GE plant to become an agricultural weed, or to cause damage to agriculture<br />
through the introduction <strong>of</strong> a novel plant pathogen produced by the transgenic plant or a change in plant susceptibility to pests.<br />
Under FIFRA the benefits <strong>of</strong> products as well as their potential risks are evaluated in the registration process, so the EPA assesses<br />
the potential economic impact <strong>of</strong> the introduction <strong>of</strong> the PIP product, along with the environmental <strong>and</strong> human health benefits <strong>of</strong><br />
the altered pesticide-use regime. When appropriate, the EPA evaluates insect resistance management (IRM) plans proposed<br />
by applicants to confirm that the plan will be sufficient to delay resistance development to Bt. Both EPA <strong>and</strong> USDA-APHIS are<br />
concerned with the potential for gene flow to occur from the transgenic plant to wild relatives (Table 1). In the case <strong>of</strong> plants<br />
engineered to produce a PIP, USDA-APHIS <strong>and</strong> EPA evaluate whether there will be toxicity to non-target organisms that might<br />
come into contact with the crop or its residues.<br />
Using science for risk management: the insect-resistance management example<br />
In addition to the data provided to the agencies during their assessment <strong>of</strong> specific products during the approval process, both the<br />
EPA <strong>and</strong> USDA fund active research programs to continue studying the environmental <strong>and</strong> human health impacts <strong>of</strong> GE crop<br />
plants. An example <strong>of</strong> the use <strong>of</strong> science in determining regulatory policy is that <strong>of</strong> the EPA’s IRM plan for PIPs utilizing proteins<br />
produced by the bacterium Bacillus thuringiensis (Bt), which are the most common PIPs engineered into plants.<br />
Insect populations exposed to pesticides over a long enough timeframe<br />
will develop resistance (Feyereisen 1995), so because preparations<br />
<strong>of</strong> the bacteria that express Bt toxins are an important pest<br />
management tool for the organic farming industry, concerns were<br />
raised that the development <strong>of</strong> resistance to Bt would deprive the agricultural<br />
community <strong>of</strong> a safe, environmentally friendly pesticide. Due<br />
to the adverse health <strong>and</strong> environmental effects <strong>of</strong> having to use conventional<br />
pesticides instead <strong>of</strong> Bt, the EPA has required a very stringent<br />
IRM plan for the use <strong>of</strong> Bt-PIP-containing crops to delay resistance<br />
development, in contrast to almost all other pesticides.<br />
After studying insect-resistance models <strong>and</strong> experimental data, the<br />
80 : 20 external<br />
refuge<br />
(a)<br />
80 : 20 external<br />
refuge<br />
(b)<br />
50 : 50 refuge in<br />
cotton growing areas<br />
95 : 5 embedded<br />
refuge<br />
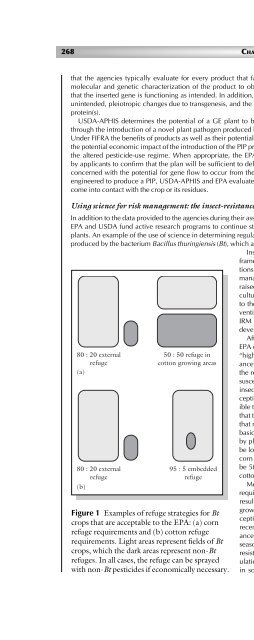
Figure 1 Examples <strong>of</strong> refuge strategies for Bt<br />
crops that are acceptable to the EPA: (a) corn<br />
refuge requirements <strong>and</strong> (b) cotton refuge<br />
requirements. Light areas represent fields <strong>of</strong> Bt<br />
crops, which the dark areas represent non-Bt<br />
refuges. In all cases, the refuge can be sprayed<br />
with non-Bt pesticides if economically necessary.<br />
EPA developed a program to delay resistance development based on a<br />
“high dose/structured refuge” approach. This strategy relies on resistance<br />
to Bt being a genetically recessive trait <strong>and</strong> the initial frequency <strong>of</strong><br />
the resistance allele being very low. When this is the case, refuges for<br />
susceptible insects can be designed so that in principle any resistant<br />
insects that arise in the population will almost certainly mate with a susceptible<br />
individual so that the heterozygous <strong>of</strong>fspring will be susceptible<br />
to the PIP. The high dose requirement for PIP products necessitates<br />
that the plant expresses a level <strong>of</strong> Bt protein at least 25-fold greater than<br />
that needed to kill 99% <strong>of</strong> susceptible insects in laboratory assays. The<br />
basic structured refuge requirements for Bt crops are satisfied in general<br />
by planting 20% <strong>of</strong> the field as a contiguous non-Bt refuge that should<br />
be located within 0.8 km <strong>of</strong> the Bt crop fields (Figure 1). However, if Bt<br />
corn is planted in cotton-producing areas then the non-Bt refuge should<br />
be 50% <strong>of</strong> the corn acreage because cotton pests could feed on both<br />
cotton <strong>and</strong> corn <strong>and</strong> develop resistance more rapidly.<br />
Monitoring for insect-resistance development has always been a<br />
requirement by the EPA for registrants, who inform the EPA <strong>of</strong> the<br />
results <strong>of</strong> their monitoring program on an annual basis, along with any<br />
grower observations <strong>of</strong> increased crop damage by insects normally susceptible<br />
to Bt toxins. In addition, academic researchers have performed<br />
recent studies in Arizona, North Carolina, <strong>and</strong> Iowa to measure resistance<br />
development in Bt corn <strong>and</strong> cotton fields over multiple growing<br />
seasons (Tabashnik et al. 2003). In most cases, the initial frequency <strong>of</strong><br />
resistance alleles to particular GE Bt-containing plants in the target populations<br />
was very low (