- Page 2 and 3:
Industrial Biotransformations Edite
- Page 4 and 5:
Industrial Biotransformations Secon
- Page 6 and 7:
Contents Preface to the first editi
- Page 8 and 9:
5 Basics of Bioreaction Engineering
- Page 10 and 11:
X Preface to the first edition many
- Page 12 and 13:
XII Preface to the second edition E
- Page 14 and 15:
XIV List of Contributors Prof. Dr.
- Page 16 and 17:
2 1 History of Industrial Biotransf
- Page 18 and 19:
4 1 History of Industrial Biotransf
- Page 20 and 21:
6 1 History of Industrial Biotransf
- Page 22 and 23:
8 1 History of Industrial Biotransf
- Page 24 and 25:
10 1 History of Industrial Biotrans
- Page 26 and 27:
12 1 History of Industrial Biotrans
- Page 28 and 29:
14 1 History of Industrial Biotrans
- Page 30 and 31:
R' 16 O H N 1 History of Industrial
- Page 32 and 33:
18 1 History of Industrial Biotrans
- Page 34 and 35:
20 1 History of Industrial Biotrans
- Page 36 and 37:
22 1 History of Industrial Biotrans
- Page 38 and 39:
24 1 History of Industrial Biotrans
- Page 40 and 41:
26 1 History of Industrial Biotrans
- Page 42 and 43:
28 1 History of Industrial Biotrans
- Page 44 and 45:
30 1 History of Industrial Biotrans
- Page 46 and 47:
32 1 History of Industrial Biotrans
- Page 48 and 49:
34 1 History of Industrial Biotrans
- Page 50 and 51:
36 1 History of Industrial Biotrans
- Page 52 and 53:
38 2 The Enzyme Classification Tab.
- Page 54 and 55:
40 2 The Enzyme Classification trib
- Page 56 and 57:
42 2 The Enzyme Classification EC 1
- Page 58 and 59:
44 2 The Enzyme Classification EC 1
- Page 60 and 61:
46 2 The Enzyme Classification EC 1
- Page 62 and 63:
48 2 The Enzyme Classification EC 2
- Page 64 and 65:
50 2 The Enzyme Classification EC 3
- Page 66 and 67:
52 2 The Enzyme Classification EC 3
- Page 68 and 69:
54 2 The Enzyme Classification 2.2.
- Page 70 and 71:
56 2 The Enzyme Classification The
- Page 72 and 73:
58 2 The Enzyme Classification EC 5
- Page 74 and 75:
60 2 The Enzyme Classification in g
- Page 76 and 77:
62 2 The Enzyme Classification Refe
- Page 78 and 79:
64 3 Retrosynthetic Biocatalysis 3.
- Page 80 and 81:
66 3 Retrosynthetic Biocatalysis R
- Page 82 and 83:
68 3 Retrosynthetic Biocatalysis 3.
- Page 84 and 85:
70 3 Retrosynthetic Biocatalysis 3.
- Page 86 and 87:
72 3 Retrosynthetic Biocatalysis 3.
- Page 88 and 89:
74 3 Retrosynthetic Biocatalysis 3.
- Page 90 and 91:
76 3 Retrosynthetic Biocatalysis R
- Page 92 and 93:
78 3 Retrosynthetic Biocatalysis Wh
- Page 94 and 95:
80 3 Retrosynthetic Biocatalysis 3.
- Page 96 and 97:
82 3 Retrosynthetic Biocatalysis 3.
- Page 98 and 99:
84 3 Retrosynthetic Biocatalysis 3.
- Page 100 and 101:
86 3 Retrosynthetic Biocatalysis 3.
- Page 102 and 103:
88 3 Retrosynthetic Biocatalysis Re
- Page 104 and 105:
90 3 Retrosynthetic Biocatalysis (D
- Page 106 and 107:
4 Optimization of Industrial Enzyme
- Page 108 and 109:
4.2 Learning from Nature Nature its
- Page 110 and 111:
4.3 Enzyme Production Using Bacteri
- Page 112 and 113:
4.4 Improvements to Enzymes by Mole
- Page 114 and 115:
1 4.4 Improvements to Enzymes by Mo
- Page 116 and 117:
4.4 Improvements to Enzymes by Mole
- Page 118 and 119:
4.5 Identification of Improved Enzy
- Page 120 and 121:
4.5 Identification of Improved Enzy
- Page 122 and 123:
Galleron, N., Ghim, S.Y., Glaser, P
- Page 124 and 125:
ment, Relaxation, and Reversal of t
- Page 126 and 127:
81 Goddard, J.-P., Reymond, J.-L. 2
- Page 128 and 129:
5 Basics of Bioreaction Engineering
- Page 130 and 131:
where r p = selectivity to componen
- Page 132 and 133:
5.1 Definitions operoxidase (CPO) h
- Page 134 and 135:
5.1 Definitions The residence time
- Page 136 and 137:
5.1 Definitions In iso-electric pre
- Page 138 and 139:
5.2 Biocatalyst Kinetics formation
- Page 140 and 141:
5.2 Biocatalyst Kinetics K M values
- Page 142 and 143:
v ˆ V max ‰SŠ KM 1‡ ‰PŠ KI
- Page 144 and 145:
5.3 Basic Reactor Types and their M
- Page 146 and 147:
dx concentration [S] 0 [S] 1 [S] e
- Page 148 and 149:
5.4 Biocatalyst Recycling and Recov
- Page 150 and 151:
. better stability, especially towa
- Page 152 and 153:
5.4.2 Cross-linking 5.4 Biocatalyst
- Page 154 and 155:
. immobilization methods . substrat
- Page 156 and 157:
References Owing to the need for st
- Page 158 and 159:
40 Vuorilehto, K., Lütz, S., Wandr
- Page 160 and 161:
Common name of enzyme Name of strai
- Page 162 and 163:
Common name of enzyme Name of strai
- Page 164 and 165:
Alcohol dehydrogenase Neurospora cr
- Page 166 and 167:
Alcohol dehydrogenase Neurospora cr
- Page 168 and 169:
Alcohol dehydrogenase Rhodococcus e
- Page 170 and 171:
Alcohol dehydrogenase Rhodococcus e
- Page 172 and 173:
Alcohol dehydrogenase Acinetobacter
- Page 174 and 175:
Alcohol dehydrogenase Acinetobacter
- Page 176 and 177:
Dehydrogenase Zygosaccharomyces rou
- Page 178 and 179:
Alcohol dehydrogenase Lactobacillus
- Page 180 and 181:
Alcohol dehydrogenase Lactobacillus
- Page 182 and 183:
Alcohol dehydrogenase Lactobacillus
- Page 184 and 185:
Carbonyl reductase Escherichia coli
- Page 186 and 187:
Aldehyde reductase Escherichia coli
- Page 188 and 189:
Lactate dehydrogenase Staphylococcu
- Page 190 and 191:
d-Lactate dehydrogenase Leuconostoc
- Page 192 and 193:
d-Lactate dehydrogenase Leuconostoc
- Page 194 and 195:
d-Sorbitol dehydrogenase Gluconobac
- Page 196 and 197:
d-Sorbitol dehydrogenase Gluconobac
- Page 198 and 199:
Dehydrogenase Geotrichum candidum 1
- Page 200 and 201:
Ketoreductase 1 = ethyl-4-chloro-3-
- Page 202 and 203:
Dehydrogenase Candida sorbophila N
- Page 204 and 205:
Dehydrogenase Candida sorbophila N
- Page 206 and 207:
Reductase Pichia methanolica 1 = Et
- Page 208 and 209:
Reductase Aureobasidium pullulans S
- Page 210 and 211:
Reductase Nocardia salmonicolor SC
- Page 212 and 213:
Glutamate dehydrogenase / Glucose 1
- Page 214 and 215:
Leucine dehydrogenase Bacillus spha
- Page 216 and 217:
Leucine dehydrogenase Bacillus spha
- Page 218 and 219:
Phenylalanine dehydrogenase / Forma
- Page 220 and 221:
d-Aminoacid oxidase Trijonopsis var
- Page 222 and 223:
d-Amino acid oxidase Trigonopsis va
- Page 224 and 225:
Nicotinic acid hydroxylase Achromob
- Page 226 and 227:
Nicotinic acid hydroxylase Achromob
- Page 228 and 229:
Catalase Microbial source 1) Reacti
- Page 230 and 231:
Oxygenase Arthrobacter sp. 1) React
- Page 232 and 233:
Naphthalene dioxygenase Pseudomonas
- Page 234 and 235:
Naphthalene dioxygenase Pseudomonas
- Page 236 and 237:
Benzoate dioxygenase Pseudomonas pu
- Page 238 and 239:
Cyclohexanone monooxygenase Acineto
- Page 240 and 241:
Cyclohexanone monooxygenase Acineto
- Page 242 and 243:
Oxygenase Escherichia coli OH OH 1
- Page 244 and 245:
Monooxygenases / Aryl alcohol dehyd
- Page 246 and 247:
Styrene monooxygenase Escherichia c
- Page 248 and 249:
Styrene monooxygenase Escherichia c
- Page 250 and 251:
Monooxygenase Pseudomonas putida H
- Page 252 and 253:
Monooxygenase Streptomyces sp. SC 1
- Page 254 and 255:
Oxygenase Nocardia autotropica 1) R
- Page 256 and 257:
Monooxygenase Nocardia corallina 1
- Page 258 and 259:
Monooxygenase Nocardia corallina
- Page 260 and 261:
Monooxygenase Nocardia corallina O
- Page 262 and 263:
Oxidase Pseudomonas oleovorans 1) R
- Page 264 and 265:
Reductase Baker’s yeast 1 = oxois
- Page 266 and 267:
Oxidase Rhodococcus erythropolis 2
- Page 268 and 269:
Desaturase Rhodococcus sp. 1) React
- Page 270 and 271:
Desaturase Rhodococcus sp. 3) Flow
- Page 272 and 273:
Oxidase Beauveria bassiana 1) React
- Page 274 and 275:
Oxidase Beauveria bassiana ● The
- Page 276 and 277:
Cyclodextrin glycosyltransferase Ba
- Page 278 and 279:
d-Amino acid transaminase Bacillus
- Page 280 and 281:
d-Amino acid transaminase Bacillus
- Page 282 and 283:
Transaminase Bacillus megaterium 1)
- Page 284 and 285:
Lipase Burkholderia plantarii 2 (R,
- Page 286 and 287:
Lipase Burkholderia plantarii 3) Fl
- Page 288 and 289:
Lipase Pseudomonas cepacia 1 = cis-
- Page 290 and 291:
Lipase Pseudomonas cepacia 5) Produ
- Page 292 and 293:
Lipase Pseudomonas cepacia 2 F 1 =
- Page 294 and 295:
Lipase Pseudomonas cepacia 6) Liter
- Page 296 and 297:
Lipase Mucor miehei Fig. 3.1.1.3 -
- Page 298 and 299:
Lipase Mucor miehei 6) Literature E
- Page 300 and 301:
Lipase Porcine pancreas 5) Product
- Page 302 and 303:
Lipase Pseudomonas fluorescens Fig.
- Page 304 and 305:
Lipase Pseudomonas fluorescens O O
- Page 306 and 307:
Lipase Candida cylindracea ● In c
- Page 308 and 309:
Lipase Candida antarctica 2 F 1) Re
- Page 310 and 311:
Lipase Candida antarctica ● It sh
- Page 312 and 313:
Lipase Candida antarctica ● The p
- Page 314 and 315:
Lipase Candida antarctica 3) Flow s
- Page 316 and 317:
Lipase Arthrobacter sp. ● For thi
- Page 318 and 319:
Lipase Serratia marescens MeO R MeO
- Page 320 and 321:
Lipase Serratia marescens Fig. 3.1.
- Page 322 and 323:
Lipase Pseudomonas cepacia 1) React
- Page 324 and 325:
Lipase Candida antarctica 1) Reacti
- Page 326 and 327:
Lipase Candida antarctica 4) Proces
- Page 328 and 329:
-Galactosidase Saccharomyces lactis
- Page 330 and 331:
Lipase Pseudomonas cepacia ● The
- Page 332 and 333:
Lactonase Fusarium oxysporum 1 = pa
- Page 334 and 335:
Lactonase Fusarium oxysporum Fig. 3
- Page 336 and 337:
Lactonase Fusarium oxysporum 5) Pro
- Page 338 and 339:
Glutaryl amidase Escherichia coli F
- Page 340 and 341:
Glutaryl amidase Escherichia coli 6
- Page 342 and 343:
Glutaryl amidase Pseudomonas sp. 3)
- Page 344 and 345:
a-Amylase / Amyloglucosidase Bacill
- Page 346 and 347:
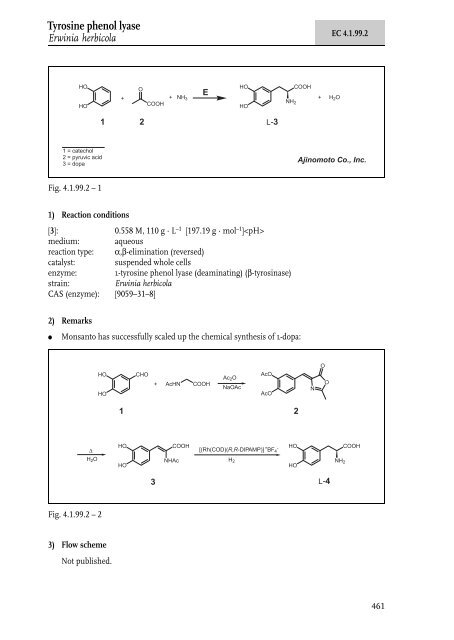
Nucleosidase / Phosphorylase Erwini
- Page 348 and 349:
Aminopeptidase Pseudomonas putida 1
- Page 350 and 351:
Aminopeptidase Pseudomonas putida
- Page 352 and 353:
Aminopeptidase Pseudomonas putida 3
- Page 354 and 355:
Aminopeptidase Pseudomonas putida
- Page 356 and 357:
Carboxypeptidase B Pig Pancreas 3)
- Page 358 and 359:
Carboxypeptidase B Pig Pancreas 2)
- Page 360 and 361:
Carboxypeptidase B Pig Pancreas 3)
- Page 362 and 363:
Trypsin Pig Pancreas 2) Remarks ●
- Page 364 and 365:
Trypsin Pig Pancreas 2) Remarks ●
- Page 366 and 367:
Trypsin Pig Pancreas 2) Remarks ●
- Page 368 and 369:
Subtilisin Bacillus licheniformis 1
- Page 370 and 371:
Subtilisin Bacillus licheniformis 4
- Page 372 and 373:
Subtilisin Bacillus licheniformis
- Page 374 and 375:
Subtilisin Bacillus licheniformis 6
- Page 376 and 377:
Subtilisin Bacillus licheniformis
- Page 378 and 379:
Subtilisin Bacillus sp. EtOOC (R/S)
- Page 380 and 381:
Subtilisin Bacillus sp. drug discov
- Page 382 and 383:
Subtilisin Bacillus lentus 1 = (R,S
- Page 384 and 385:
Thermolysin Bacillus thermoproteoly
- Page 386 and 387:
Thermolysin Bacillus thermoproteoly
- Page 388 and 389:
Amidase Comamonas acidovorans 1) Re
- Page 390 and 391:
Amidase Klebsiella terrigena 2 H N
- Page 392 and 393:
Amidase Klebsiella terrigena 6) Lit
- Page 394 and 395:
Amidase Klebsiella oxytoca company:
- Page 396 and 397:
Urease Lactobacillus fermentum ●
- Page 398 and 399:
Penicillin amidase Escherichia coli
- Page 400 and 401:
Penicillin amidase Escherichia coli
- Page 402 and 403:
Penicillin amidase Bacillus megater
- Page 404 and 405:
Penicillin amidase Escherichia coli
- Page 406 and 407:
Penicillin amidase Escherichia coli
- Page 408 and 409:
Penicillin acylase Escherichia coli
- Page 410 and 411:
Penicillin acylase Escherichia coli
- Page 412 and 413:
Penicillin acylase Escherichia coli
- Page 414 and 415:
Aminoacylase Aspergillus niger 2 N
- Page 416 and 417:
Aminoacylase Aspergillus niger 3) F
- Page 418 and 419:
Aminoacylase Aspergillus oryzae S C
- Page 420 and 421:
Aminoacylase Aspergillus oryzae 3)
- Page 422 and 423: d-Hydantoinase Bacillus brevis Fig.
- Page 424 and 425: d-Hydantoinase Bacillus brevis 3) F
- Page 426 and 427: Hydantoinase / Carbamoylase Pseudom
- Page 428 and 429: l-Hydantoinase Arthrobacter sp. DSM
- Page 430 and 431: l-Hydantoinase Arthrobacter sp. DSM
- Page 432 and 433: -Lactamase Aureobacterium sp. 3) Fl
- Page 434 and 435: -Lactamase Pseudomonas solanacearum
- Page 436 and 437: Lactamase / Racemase Cryptococcus l
- Page 438 and 439: Nitrilase Acidovorax facilis 1 = 2-
- Page 440 and 441: Nitrilase Escherichia coli 1) React
- Page 442 and 443: Nitrilase / Hydroxylase Agrobacteri
- Page 444 and 445: Nitrilase / Hydroxylase Alcaligenes
- Page 446 and 447: Dehalogenase Pseudomonas putida 1)
- Page 448 and 449: Dehalogenase Pseudomonas putida 5)
- Page 450 and 451: Haloalkane dehalogenase Alcaligenes
- Page 452 and 453: Haloalkane dehalogenase Alcaligenes
- Page 454 and 455: Haloalkane dehalogenase Enterobacte
- Page 456 and 457: Halohydrin dehalogenase 1 = ethyl-(
- Page 458 and 459: Pyruvate decarboxylase Saccharomyce
- Page 460 and 461: Acetolactate decarboxylase Bacillus
- Page 462 and 463: Aspartate b-decarboxylase Pseudomon
- Page 464 and 465: Aspartate b-decarboxylase Pseudomon
- Page 466 and 467: Oxynitrilase Hevea brasiliensis 1 =
- Page 468 and 469: N-Acetyl-d-neuraminic acid aldolase
- Page 470 and 471: N-Acetyl-d-neuraminic acid aldolase
- Page 474 and 475: Fumarase Corynebacterium glutamicum
- Page 476 and 477: Fumarase Corynebacterium glutamicum
- Page 478 and 479: Fumarase Brevibacterium flavum 4) P
- Page 480 and 481: Enoyl-CoA hydratase Candida rugosa
- Page 482 and 483: Enoyl-CoA hydratase Candida rugosa
- Page 484 and 485: Tryptophan synthase Escherichia col
- Page 486 and 487: Malease Pseudomonas pseudoalcaligen
- Page 488 and 489: Malease Pseudomonas pseudoalcaligen
- Page 490 and 491: Nitrile hydratase Pseudomonas chlor
- Page 492 and 493: Nitrile hydratase Rhodococcus rhodo
- Page 494 and 495: Nitrile hydratase Rhodococcus rhodo
- Page 496 and 497: Nitrile hydratase Rhodococcus rhodo
- Page 498 and 499: Nitrile hydratase Rhodococcus rhodo
- Page 500 and 501: Carnitine dehydratase Escherichia c
- Page 502 and 503: Carnitine dehydratase Escherichia c
- Page 504 and 505: Carnitine dehydratase Escherichia c
- Page 506 and 507: Aspartase Escherichia coli 3) Flow
- Page 508 and 509: Aspartase Escherichia coli 5) Produ
- Page 510 and 511: Aspartase Brevibacterium flavum 3)
- Page 512 and 513: l-Aspartase Escherichia coli 3) Flo
- Page 514 and 515: l-Phenylalanine ammonia-lyase Rhodo
- Page 516 and 517: Amino acid racemase Amycolatopsis o
- Page 518 and 519: GlcNAc 2-epimerase Escherichia coli
- Page 520 and 521: Xylose isomerase Bacillus coagulans
- Page 522 and 523:
Xylose isomerase Bacillus coagulans
- Page 524 and 525:
a-Glucosyl transferase Protaminobac
- Page 526 and 527:
516 7 Quantitative Analysis of Indu
- Page 528 and 529:
518 7 Quantitative Analysis of Indu
- Page 530 and 531:
520 7 Quantitative Analysis of Indu
- Page 532 and 533:
522 Index of enzyme name enzyme nam
- Page 534 and 535:
524 Index of enzyme name enzyme nam
- Page 536 and 537:
526 Index of strain strain enzyme n
- Page 538 and 539:
528 Index of strain strain enzyme n
- Page 540 and 541:
530 Index of strain strain enzyme n
- Page 542 and 543:
532 Index of company company strain
- Page 544 and 545:
534 Index of company company strain
- Page 546 and 547:
536 Index of starting material star
- Page 548 and 549:
538 Index of starting material star
- Page 550 and 551:
540 Index of starting material star
- Page 552 and 553:
542 Index of starting material star
- Page 554 and 555:
544 Index of starting material star
- Page 556 and 557:
546 Index of product product enzyme
- Page 558 and 559:
548 Index of product product enzyme
- Page 560 and 561:
550 Index of product product enzyme
- Page 562 and 563:
552 Index of product product enzyme
- Page 564 and 565:
554 Index of product product enzyme
- Page 566:
556 Index of product product enzyme