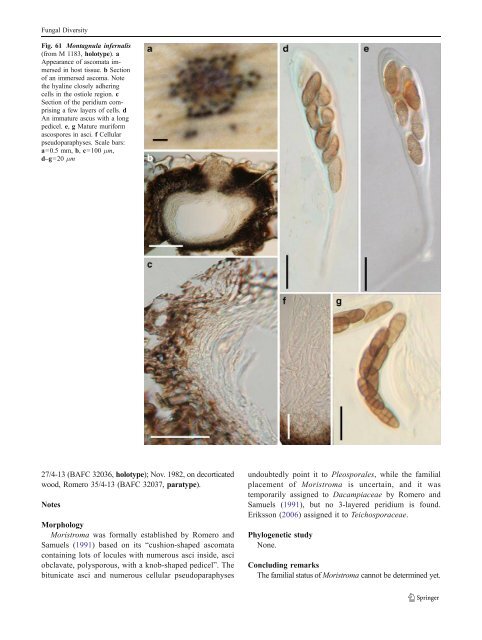
Fungal Diversity pseudoparaphyses (Fig. 61c). Hamathecium of dense, narrow cellular pseudoparaphyses, 2–4.5 μm broad, septate (Fig. 61f). Asci 153–170(−200)×17.5–21.5 μm (including pedicel), bitunicate, fissitunicate, cylindro-clavate to clavate, pedicel 28–60(−85) μm long, 8-spored, biseriate, with an ocular chamber best seen in immature ascus (to 3 μm wide×3 μm high) (Fig. 61d and e). Ascospores 24–29×9–11 μm, oblong to narrowly oblong, straight or somewhat curved, reddish brown to dark yellowish brown, verruculose, with five transverse septa and one vertical septum in each middle cells, constricted at the primary and secondary primary septa (Fig. 61g). Anamorph: none reported. Material examined: PORTUGAL, Coimbra Lusitania, on leaves of Fourcroya longava pr., Feb., 1881, leg. Moller. (M 1183, holotype). Notes Morphology Montagnula was introduced to accommodate two Pleospora species, i.e. P. infernalis (Niessl) Wehm. and P. gigantea Mont. by Berlese (1896), based on the presence of hyphal stromatic tissues over the ascomata and asci with relatively long pedicels (Barr 2001). Montagnula infernalis was selected as the lectotype species (Clements and Shear 1931). Subsequently, Wehmeyer (1957, 1961) treated Montagnula as a subgenus of Pleospora. Crivelli(1983) accepted Montagnula as a separate genus, and divided it into two subgenera, i.e. Montagnula and Rubiginospora. Montagnula was characterized by having dark brown ascospores and exclusively occurring on Agavaceae, while Rubiginospora has reddish brown ascospores and occurs on Poaceae. This proposal was not accepted by many workers (Barr 2001). Subsequently, more species with various ascospores (such as phragmosporous species by Leuchtmann (1984) and didymosporous species by Aptroot (1995) were added in this genus), which has obviously become heterogenic. Barr (2001) assigned species of Montagnula into different genera, i.e. Kalmusia and Didymosphaerella, respectively and introduced Montagnulaceae to accommodate all of these genera. Phylogenetic study Montagnula opulenta forms a robust phylogenetic clade with species of Bimuria, Curreya, Didymocrea, Letendraea, Paraphaeosphaeria, Phaeodothis and Karstenula, which might represent a familial group (Schoch et al. 2006; Zhang et al. 2009a). A more convincing conclusion can only be obtained following sequence data from more verified fungi being added to the phylogenetic tree. Concluding remarks One striking character of Montagnula infernalis is the very long ascal pedicel once it is released from the ascomata. However, this character appears to have evolved more than once and can be found in Kirschsteiniothelia elaterascus Shearer which clusters with Helicascus (Shearer et al. 2009). The same ascus character is also found in Xenolophium and Ostropella in the Platystomaceae (Mugambi and Huhndorf 2009b). Montagnula opulenta is a didymosporous species, but phylogenetically closely related to those dictyosporous (Karstenula rhodostoma) and phragmosporous (Paraphaeosphaeria michotii) members of Montagnulaceae (Zhang et al. 2009a). This might indicate that compared to other morphological characters, ascospore type is not a valid character at family level classification. Moristroma A.I. Romero & Samuels, Sydowia 43: 246 (1991). (<strong>Pleosporales</strong>, genera incertae sedis) Generic description Habitat terrestrial, saprobic. Ascomata medium-sized, solitary, scattered, or in small groups, superficial, cushion-like, circular in outline, wall black, roughened, containing numerous locules. Peridium thin, 1-layered. Hamathecium of dense, long filliform pseudoparaphyses, 2–3 μm broad, septate, branching. Asci polysporous, with a short, laterally displaced, sometimes papillate knob-shaped pedicel, apex very thick walled, bitunicate, fissitunicate, obclavate, ocular chamber not observed. Polyspores oblong to cylindrical, hyaline, non-septate. Anamorphs reported for genus: none. Literature: Eriksson 2006; Romero and Samuels 1991. Type species Moristroma polysporum A.I. Romero & Samuels, Sydowia 43: 246 (1991). (Fig. 62) Ascomata 100–210 μm high×340–600 μm diam., solitary, scattered, or in small groups of 2–3, superficial, with basal wall remaining immersed in host tissue, cushion-like, circular in outline, wall black, roughened, containing numerous locules, each locule 120–240 μm diam., ostiolate (Fig. 62a and b). Peridium 14–30 μm thick, 1-layered, composed of small heavily pigmented thick-walled cells of textura angularis, cells2–4 μm diam., cell wall 1.5–3 μm thick, peridium between the locules hyaline (Fig. 62b and c). Hamathecium of dense, long filliform pseudoparaphyses, 2– 3 μm broad, septate, branching. Asci 44–60×12–14 μm (x ¼ 54:3 13mm, n=10), polysporous, with a short, papillate knob-shaped pedicel, apex very thick-walled, bitunicate, fissitunicate, obclavate, ocular chamber not observed (Fig. 62d and e). Polyspores 3–4(−5)×0.6–1.2 μm, oblong to cylindrical, hyaline, non-septate, smooth (Fig. 62f). Anamorph: none reported. Material examined: ARGENTINA, Buenos Aires, Ramallo, on Eucalyptus viminalis Labill., May 1982, Romero
Fungal Diversity Fig. 61 Montagnula infernalis (from M 1183, holotype). a Appearance of ascomata immersed in host tissue. b Section of an immersed ascoma. Note the hyaline closely adhering cells in the ostiole region. c Section of the peridium comprising a few layers of cells. d An immature ascus with a long pedicel. e, g Mature muriform ascospores in asci. f Cellular pseudoparaphyses. Scale bars: a=0.5 mm, b, c=100 μm, d–g=20 μm 27/4-13 (BAFC 32036, holotype); Nov. 1982, on decorticated wood, Romero 35/4-13 (BAFC 32037, paratype). Notes Morphology Moristroma was formally established by Romero and Samuels (1991) based on its “cushion-shaped ascomata containing lots of locules with numerous asci inside, asci obclavate, polysporous, with a knob-shaped pedicel”. The bitunicate asci and numerous cellular pseudoparaphyses undoubtedly point it to <strong>Pleosporales</strong>, while the familial placement of Moristroma is uncertain, and it was temporarily assigned to Dacampiaceae by Romero and Samuels (1991), but no 3-layered peridium is found. Eriksson (2006) assignedittoTeichosporaceae. Phylogenetic study None. Concluding remarks The familial status of Moristroma cannot be determined yet.
- Page 1 and 2:
Fungal Diversity DOI 10.1007/s13225
- Page 3 and 4:
Fungal Diversity Table 1 Major circ
- Page 5 and 6:
Fungal Diversity
- Page 7 and 8:
Fungal Diversity biocontrol agent o
- Page 9 and 10:
Fungal Diversity substrates and man
- Page 11 and 12:
Fungal Diversity 2. To investigate
- Page 13 and 14:
Fungal Diversity Table 3 (continued
- Page 15 and 16:
Fungal Diversity Table 3 (continued
- Page 17 and 18:
Fungal Diversity Table 3 (continued
- Page 19 and 20:
Fungal Diversity
- Page 21 and 22:
Fungal Diversity Fig. 2 Aigialus gr
- Page 23 and 24:
Fungal Diversity Fig. 3 Amniculicol
- Page 25 and 26:
Fungal Diversity Literature: Berkel
- Page 27 and 28:
Fungal Diversity Ascorhombispora L.
- Page 29 and 30:
Fungal Diversity
- Page 31 and 32:
Fungal Diversity Fig. 8 Astrosphaer
- Page 33 and 34:
Fungal Diversity Fig. 9 Asymmetrico
- Page 35 and 36:
Fungal Diversity Notes Morphology B
- Page 37 and 38:
Fungal Diversity Generic descriptio
- Page 39 and 40:
Fungal Diversity Anamorph: none rep
- Page 41 and 42:
Fungal Diversity Fig. 14 Bimuria no
- Page 43 and 44:
Fungal Diversity Fig. 15 Bricookea
- Page 45 and 46:
Fungal Diversity Fig. 16 Byssolophi
- Page 47 and 48:
Fungal Diversity Notes Morphology B
- Page 49 and 50:
Fungal Diversity the reaction of pe
- Page 51 and 52:
Fungal Diversity
- Page 53 and 54:
Fungal Diversity Fig. 21 Chaetomast
- Page 55 and 56:
Fungal Diversity
- Page 57 and 58:
Fungal Diversity Fig. 23 Cilioplea
- Page 59 and 60:
Fungal Diversity with one or two ve
- Page 61 and 62:
Fungal Diversity Moreau 1953; Munk
- Page 63 and 64:
Fungal Diversity Material examined:
- Page 65 and 66:
Fungal Diversity Fig. 28 Dothidotth
- Page 67 and 68:
Fungal Diversity Fig. 29 Dubitatio
- Page 69 and 70: Fungal Diversity assigned Entodesmi
- Page 71 and 72: Fungal Diversity fusoid to somewhat
- Page 73 and 74: Fungal Diversity Fig. 33 Hadrospora
- Page 75 and 76: Fungal Diversity Fig. 34 Halotthia
- Page 77 and 78: Fungal Diversity Notes Morphology H
- Page 79 and 80: Fungal Diversity some effused Hypox
- Page 81 and 82: Fungal Diversity Fig. 38 Isthmospor
- Page 83 and 84: Fungal Diversity Fig. 39 Kalmusia e
- Page 85 and 86: Fungal Diversity ascospores were br
- Page 87 and 88: Fungal Diversity furcate pedicel an
- Page 89 and 90: Fungal Diversity Anamorph: none rep
- Page 91 and 92: Fungal Diversity
- Page 93 and 94: Fungal Diversity Material examined:
- Page 95 and 96: Fungal Diversity Fig. 46 Lewia scro
- Page 97 and 98: Fungal Diversity Fig. 47 Lichenopyr
- Page 99 and 100: Fungal Diversity Loculohypoxylon M.
- Page 101 and 102: Fungal Diversity cells small heavil
- Page 103 and 104: Fungal Diversity upper place, septa
- Page 105 and 106: Fungal Diversity
- Page 107 and 108: Fungal Diversity (CBS 627.86) was i
- Page 109 and 110: Fungal Diversity Fig. 54 Mamillisph
- Page 111 and 112: Fungal Diversity Fig. 55 Massarina
- Page 113 and 114: Fungal Diversity phaeria as a synon
- Page 115 and 116: Fungal Diversity 5-8 μm diam., ind
- Page 117 and 118: Fungal Diversity cell wall
- Page 119: Fungal Diversity Fig. 60 Mixtura sa
- Page 123 and 124: Fungal Diversity spored, bitunicate
- Page 125 and 126: Fungal Diversity Fig. 64 Murispora
- Page 127 and 128: Fungal Diversity Type species Neoph
- Page 129 and 130: Fungal Diversity brown, 8-septate,
- Page 131 and 132: Fungal Diversity Fig. 68 Ohleria mo
- Page 133 and 134: Fungal Diversity Fig. 69 Ohleriella
- Page 135 and 136: Fungal Diversity Fig. 70 Ophiobolus
- Page 137 and 138: Fungal Diversity Type species Ostro
- Page 139 and 140: Fungal Diversity
- Page 141 and 142: Fungal Diversity (Shoemaker and Bab
- Page 143 and 144: Fungal Diversity ium thin, composed
- Page 145 and 146: Fungal Diversity Fig. 76 Platysporo
- Page 147 and 148: Fungal Diversity Fig. 77 1 Pleomass
- Page 149 and 150: Fungal Diversity Fig. 78 Pleophragm
- Page 151 and 152: Fungal Diversity papillate, ostiola
- Page 153 and 154: Fungal Diversity Williams 1963; Mal
- Page 155 and 156: Fungal Diversity Generic descriptio
- Page 157 and 158: Fungal Diversity composed of one ce
- Page 159 and 160: Fungal Diversity Fig. 84 Saccharico
- Page 161 and 162: Fungal Diversity and nearly black a
- Page 163 and 164: Fungal Diversity dense, long trabec
- Page 165 and 166: Fungal Diversity
- Page 167 and 168: Fungal Diversity
- Page 169 and 170: Fungal Diversity Anamorphs reported
- Page 171 and 172:
Fungal Diversity
- Page 173 and 174:
Fungal Diversity
- Page 175 and 176:
Fungal Diversity Fig. 94 Westerdyke
- Page 177 and 178:
Fungal Diversity Fig. 95 Wettsteini
- Page 179 and 180:
Fungal Diversity Fig. 96 Wilmia bra
- Page 181 and 182:
Fungal Diversity Current name: Astr
- Page 183 and 184:
Fungal Diversity spores are actuall
- Page 185 and 186:
Fungal Diversity Fig. 100 Sporormie
- Page 187 and 188:
Fungal Diversity
- Page 189 and 190:
Fungal Diversity Fig. 102 Kriegerie
- Page 191 and 192:
Fungal Diversity Phylogenetic study
- Page 193 and 194:
Fungal Diversity Fig. 104 Zeuctomor
- Page 195 and 196:
Fungal Diversity Fig. 105 Muroia ni
- Page 197 and 198:
Fungal Diversity pseudoparenchymato
- Page 199 and 200:
Fungal Diversity Eremodothis Arx, K
- Page 201 and 202:
Fungal Diversity Type species: Macr
- Page 203 and 204:
Fungal Diversity ascospores of Plat
- Page 205 and 206:
Fungal Diversity monoceras Alcorn n
- Page 207 and 208:
Fungal Diversity tomataceae, Melano
- Page 209 and 210:
Fungal Diversity Table 4 (continued
- Page 211 and 212:
Fungal Diversity 1987b). Based on a
- Page 213 and 214:
Fungal Diversity only do so under v
- Page 215 and 216:
Fungal Diversity Dennis RWG (1968)
- Page 217 and 218:
Fungal Diversity Kirk PM, Cannon PF
- Page 219 and 220:
Fungal Diversity Saccardo PA (1880)
- Page 221:
Fungal Diversity Winter G (1887) As