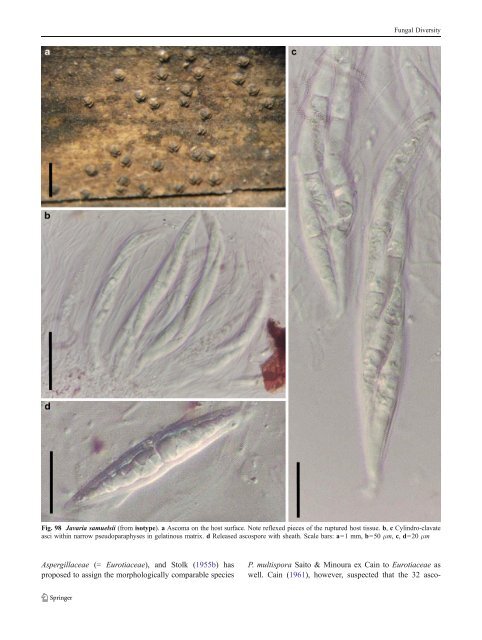
Fungal Diversity Fig. 98 Javaria samuelsii (from isotype). a Ascoma on the host surface. Note reflexed pieces of the ruptured host tissue. b, c Cylindro-clavate asci within narrow pseudoparaphyses in gelatinous matrix. d Released ascospore with sheath. Scale bars: a=1 mm, b=50 μm, c, d=20 μm Aspergillaceae (= Eurotiaceae), and Stolk (1955b) has proposed to assign the morphologically comparable species P. multispora Saito & Minoura ex Cain to Eurotiaceae as well. Cain (1961), however, suspected that the 32 asco-
Fungal Diversity spores are actually the disarticulated segments of eight 4- celled ascospores, thus assigned it under Preussia (Sporormiaceae). After detailed study, Thompson and Backus (1966) confirmed that the so-called “eight 4-celled ascospores” do not exist in the development of the asci in both P. dispersa and P. multisporum. Thus, Pycnidiophora was assigned to Eurotiaceae (Eurotiales) (Thompson and Backus 1966). Phylogenetic study Phylogenetic study based on the ITS-nLSU rDNA sequences indicated that Pycnidiophora dispersa nested within clade of Westerdykella (including the generic type, W. ornata) (Kruys and Wedin 2009). Morphologically, both genera have cleistothecioid ascomata, asci with short or without pedicels and ascospores 1-celled and no germ slits. Thus, Pycnidiophora is treated as a synonym of Westerdykella (Kruys and Wedin 2009). Concluding remarks Although the pleosporalean status of Pycnidiophora is verified, morphological characters such as the cleistothecioid ascomata and irregularly arranged asci, which do not show typical bitunicate or fissitunicate characters, absence of pseudoparaphyses as well as the ascospores separating into partspores very early all challenge the traditional concept of <strong>Pleosporales</strong> (Zhang et al. 2009a). Obviously, most of these morphological characters overlap with those of the Eurotiales. Sporormiella Ellis & Everh., N. Amer. Pyren.: 136 (1892). (Sporormiaceae) Current name: Preussia Fuckel, Hedwigia 6: 175 (1867) [1869–70]. Generic description Habitat terrestrial, saprobic (coprophilous). Ascomata medium-sized, solitary, scattered, or in small groups, semi-immersed to nearly superficial, globose, subglobose, black, coriaceous, ostiolate, periphysate. Peridium thin, composed of small heavily pigmented cells of textura angularis, apex cells smaller and walls thicker. Hamathecium of dense, septate, cellular pseudoparaphyses, embedded in mucilage. Asci 8-spored, bitunicate, fissitunicate, cylindro-clavate, with a narrowed, furcate pedicel. Ascospores cylindrical with rounded ends, brown, 3-septate, deeply constricted at each septa, with sigmoid germ slit in each cell. Anamorphs reported for genus: none. Literature: Ahmed and Cain 1972; Ellis and Everhart 1892; Khan and Cain 1979a, b; Luck-Allen and Cain 1975. Type species Sporormiella nigropurpurea Ellis & Everh., N. Amer. Pyren.: 136 (1892). (Fig. 100) Current name: Preussia nigropurpurea (Ellis & Everh.) Kruys, Syst. Biod. 7: 476. Ascomata 314–528 μm high×(250-)357–500 μm diam., solitary, scattered, or in small groups, immersed, semiimmersed to nearly superficial, globose, subglobose, wall black, coriaceous, smooth, papillate, papilla 43–115 μm long, 72–157 μm broad, ostiolate, ostiole filled with periphyses (Fig. 100a and b). Peridium 20–28 μm thick laterally, up to 40 μm thick at the apex, composed of small heavily pigmented cells of textura angularis, cells 5–8 μm diam., cell wall 1–3 μm thick, apex cells smaller and walls thicker (Fig. 100c). Hamathecium of dense, long, septate, cellular pseudoparaphyses, 1.5–2 μm broad, embedded in mucilage. Asci (70-)110–158×9–12.5(−15) μm (x ¼ 114:3 11:1mm, n= 10), 8-spored, bitunicate, fissitunicate, cylindrical to cylindroclavate, with a narrowed, furcate pedicel, 13–38 μm long, ocular chamber apparent (Fig. 100d and e). Ascospores 15– 20×4–5.5 μm (x ¼ 17:3 4:9mm, n=10), obliquely uniseriate and partially overlapping to biseriate, shortly cylindrical with rounded ends, brown, 3-septate, deeply constricted at each septum, with sigmoid germ slit in each cell, smoothwalled (Fig. 100f and g). Anamorph: none reported. Material examined: USA, New field, New Jersey: Gloucester Co., on cow dung, Mar. 1891 (NY, holotype). Notes Morphology Sporormiella was formally established by Ellis and Everhart (1892) based on the single species, Sporormiella nigropurpurea, which is characterized by its “immersed to semi-immersed, papillate ascomata, cylindrical to cylindroclavate asci with a pedicel, three to multi-septate ascospores with elongated germ slits through the whole cell” (Ahmed and Cain 1972; Khan and Cain 1979a, b). Barr (1990a) has indicated that Sporormiella might be a synonym of Ohleriella, while Sporormiella is assigned to Sporormiaceae as a separate genus (Eriksson 2006; Lumbsch and Huhndorf 2007). Currently, about 90 species are included in this genus (http://www.mycobank.org). Phylogenetic study The phylogenetic analysis based on ITS-nLSU rDNA, mtSSU rDNA and ß-tubulin sequences indicated that Sporormiella nested in Preussia,andaSporormiella–Preussia complex is formed (Kruys and Wedin 2009). Thus, Sporormiella was assigned under Preussia (Kruys and Wedin 2009).
- Page 1 and 2:
Fungal Diversity DOI 10.1007/s13225
- Page 3 and 4:
Fungal Diversity Table 1 Major circ
- Page 5 and 6:
Fungal Diversity
- Page 7 and 8:
Fungal Diversity biocontrol agent o
- Page 9 and 10:
Fungal Diversity substrates and man
- Page 11 and 12:
Fungal Diversity 2. To investigate
- Page 13 and 14:
Fungal Diversity Table 3 (continued
- Page 15 and 16:
Fungal Diversity Table 3 (continued
- Page 17 and 18:
Fungal Diversity Table 3 (continued
- Page 19 and 20:
Fungal Diversity
- Page 21 and 22:
Fungal Diversity Fig. 2 Aigialus gr
- Page 23 and 24:
Fungal Diversity Fig. 3 Amniculicol
- Page 25 and 26:
Fungal Diversity Literature: Berkel
- Page 27 and 28:
Fungal Diversity Ascorhombispora L.
- Page 29 and 30:
Fungal Diversity
- Page 31 and 32:
Fungal Diversity Fig. 8 Astrosphaer
- Page 33 and 34:
Fungal Diversity Fig. 9 Asymmetrico
- Page 35 and 36:
Fungal Diversity Notes Morphology B
- Page 37 and 38:
Fungal Diversity Generic descriptio
- Page 39 and 40:
Fungal Diversity Anamorph: none rep
- Page 41 and 42:
Fungal Diversity Fig. 14 Bimuria no
- Page 43 and 44:
Fungal Diversity Fig. 15 Bricookea
- Page 45 and 46:
Fungal Diversity Fig. 16 Byssolophi
- Page 47 and 48:
Fungal Diversity Notes Morphology B
- Page 49 and 50:
Fungal Diversity the reaction of pe
- Page 51 and 52:
Fungal Diversity
- Page 53 and 54:
Fungal Diversity Fig. 21 Chaetomast
- Page 55 and 56:
Fungal Diversity
- Page 57 and 58:
Fungal Diversity Fig. 23 Cilioplea
- Page 59 and 60:
Fungal Diversity with one or two ve
- Page 61 and 62:
Fungal Diversity Moreau 1953; Munk
- Page 63 and 64:
Fungal Diversity Material examined:
- Page 65 and 66:
Fungal Diversity Fig. 28 Dothidotth
- Page 67 and 68:
Fungal Diversity Fig. 29 Dubitatio
- Page 69 and 70:
Fungal Diversity assigned Entodesmi
- Page 71 and 72:
Fungal Diversity fusoid to somewhat
- Page 73 and 74:
Fungal Diversity Fig. 33 Hadrospora
- Page 75 and 76:
Fungal Diversity Fig. 34 Halotthia
- Page 77 and 78:
Fungal Diversity Notes Morphology H
- Page 79 and 80:
Fungal Diversity some effused Hypox
- Page 81 and 82:
Fungal Diversity Fig. 38 Isthmospor
- Page 83 and 84:
Fungal Diversity Fig. 39 Kalmusia e
- Page 85 and 86:
Fungal Diversity ascospores were br
- Page 87 and 88:
Fungal Diversity furcate pedicel an
- Page 89 and 90:
Fungal Diversity Anamorph: none rep
- Page 91 and 92:
Fungal Diversity
- Page 93 and 94:
Fungal Diversity Material examined:
- Page 95 and 96:
Fungal Diversity Fig. 46 Lewia scro
- Page 97 and 98:
Fungal Diversity Fig. 47 Lichenopyr
- Page 99 and 100:
Fungal Diversity Loculohypoxylon M.
- Page 101 and 102:
Fungal Diversity cells small heavil
- Page 103 and 104:
Fungal Diversity upper place, septa
- Page 105 and 106:
Fungal Diversity
- Page 107 and 108:
Fungal Diversity (CBS 627.86) was i
- Page 109 and 110:
Fungal Diversity Fig. 54 Mamillisph
- Page 111 and 112:
Fungal Diversity Fig. 55 Massarina
- Page 113 and 114:
Fungal Diversity phaeria as a synon
- Page 115 and 116:
Fungal Diversity 5-8 μm diam., ind
- Page 117 and 118:
Fungal Diversity cell wall
- Page 119 and 120:
Fungal Diversity Fig. 60 Mixtura sa
- Page 121 and 122:
Fungal Diversity Fig. 61 Montagnula
- Page 123 and 124:
Fungal Diversity spored, bitunicate
- Page 125 and 126:
Fungal Diversity Fig. 64 Murispora
- Page 127 and 128:
Fungal Diversity Type species Neoph
- Page 129 and 130:
Fungal Diversity brown, 8-septate,
- Page 131 and 132: Fungal Diversity Fig. 68 Ohleria mo
- Page 133 and 134: Fungal Diversity Fig. 69 Ohleriella
- Page 135 and 136: Fungal Diversity Fig. 70 Ophiobolus
- Page 137 and 138: Fungal Diversity Type species Ostro
- Page 139 and 140: Fungal Diversity
- Page 141 and 142: Fungal Diversity (Shoemaker and Bab
- Page 143 and 144: Fungal Diversity ium thin, composed
- Page 145 and 146: Fungal Diversity Fig. 76 Platysporo
- Page 147 and 148: Fungal Diversity Fig. 77 1 Pleomass
- Page 149 and 150: Fungal Diversity Fig. 78 Pleophragm
- Page 151 and 152: Fungal Diversity papillate, ostiola
- Page 153 and 154: Fungal Diversity Williams 1963; Mal
- Page 155 and 156: Fungal Diversity Generic descriptio
- Page 157 and 158: Fungal Diversity composed of one ce
- Page 159 and 160: Fungal Diversity Fig. 84 Saccharico
- Page 161 and 162: Fungal Diversity and nearly black a
- Page 163 and 164: Fungal Diversity dense, long trabec
- Page 165 and 166: Fungal Diversity
- Page 167 and 168: Fungal Diversity
- Page 169 and 170: Fungal Diversity Anamorphs reported
- Page 171 and 172: Fungal Diversity
- Page 173 and 174: Fungal Diversity
- Page 175 and 176: Fungal Diversity Fig. 94 Westerdyke
- Page 177 and 178: Fungal Diversity Fig. 95 Wettsteini
- Page 179 and 180: Fungal Diversity Fig. 96 Wilmia bra
- Page 181: Fungal Diversity Current name: Astr
- Page 185 and 186: Fungal Diversity Fig. 100 Sporormie
- Page 187 and 188: Fungal Diversity
- Page 189 and 190: Fungal Diversity Fig. 102 Kriegerie
- Page 191 and 192: Fungal Diversity Phylogenetic study
- Page 193 and 194: Fungal Diversity Fig. 104 Zeuctomor
- Page 195 and 196: Fungal Diversity Fig. 105 Muroia ni
- Page 197 and 198: Fungal Diversity pseudoparenchymato
- Page 199 and 200: Fungal Diversity Eremodothis Arx, K
- Page 201 and 202: Fungal Diversity Type species: Macr
- Page 203 and 204: Fungal Diversity ascospores of Plat
- Page 205 and 206: Fungal Diversity monoceras Alcorn n
- Page 207 and 208: Fungal Diversity tomataceae, Melano
- Page 209 and 210: Fungal Diversity Table 4 (continued
- Page 211 and 212: Fungal Diversity 1987b). Based on a
- Page 213 and 214: Fungal Diversity only do so under v
- Page 215 and 216: Fungal Diversity Dennis RWG (1968)
- Page 217 and 218: Fungal Diversity Kirk PM, Cannon PF
- Page 219 and 220: Fungal Diversity Saccardo PA (1880)
- Page 221: Fungal Diversity Winter G (1887) As