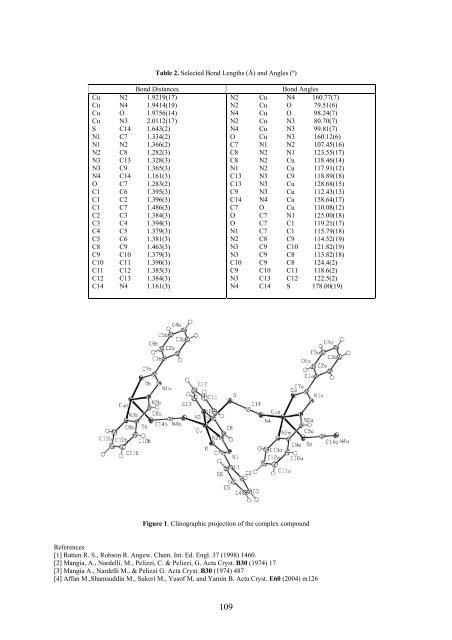
Table 2. Selected Bond Lengths (Å) and Angles (º) Bond Distances Cu N2 1.9219(17) Cu N4 1.9414(19) Cu O 1.9756(14) Cu N3 2.0112(17) S C14 1.643(2) N1 C7 1.334(2) N1 N2 1.366(2) N2 C8 1.282(3) N3 C13 1.328(3) N3 C9 1.365(3) N4 C14 1.161(3) O C7 1.283(2) C1 C6 1.395(3) C1 C2 1.396(3) C1 C7 1.486(3) C2 C3 1.384(3) C3 C4 1.394(3) C4 C5 1.379(3) C5 C6 1.381(3) C8 C9 1.463(3) C9 C10 1.379(3) C10 C11 1.390(3) C11 C12 1.385(3) C12 C13 1.384(3) C14 N4 1.161(3) Bond Angles N2 Cu N4 160.77(7) N2 Cu O 79.51(6) N4 Cu O 98.24(7) N2 Cu N3 80.70(7) N4 Cu N3 99.81(7) O Cu N3 160.12(6) C7 N1 N2 107.45(16) C8 N2 N1 123.55(17) C8 N2 Cu 118.46(14) N1 N2 Cu 117.91(12) C13 N3 C9 118.89(18) C13 N3 Cu 128.68(15) C9 N3 Cu 112.43(13) C14 N4 Cu 158.64(17) C7 O Cu 110.08(12) O C7 N1 125.00(18) O C7 C1 119.21(17) N1 C7 C1 115.79(18) N2 C8 C9 114.52(19) N3 C9 C10 121.82(19) N3 C9 C8 113.82(18) C10 C9 C8 124.4(2) C9 C10 C11 118.6(2) N3 C13 C12 122.5(2) N4 C14 S 178.00(19) Figure 1. Clinographic projection of the complex compound References [1] Ratten R. S., Robson R. Angew. Chem. Int. Ed. Engl. 37 (1998) 1460. [2] Mangia, A., Nardelli, M., Pelizzi, C. & Pelizzi, G. Acta Cryst. B30 (1974) 17 [3] Mangia A., Nardelli M., & Pelizzi G. Acta Cryst. B30 (1974) 487 [4] Affan M.,Shamsuddin M., Sukeri M., Yusof M. and Yamin B. Acta Cryst. E60 (2004) m126 109
Magnetic Phase Transition in Synthetic Cobalt-Olivine A.P. Sazonov 1* , M. Meven 2 , V. Hutanu 1 , G. Heger 1 , M. Merz 1 , V.V. Sikolenko 3 1 Institute of Cristallography, RWTH Aachen, D-52056 Aachen, Germany 2 ZWE FRM-II, TU Munich, D-85747 Garching, Germany 3 BENSC, HMI, D-14109 Berlin, Germany *E-mail: andrew.sazonov@frm2.tum.de Olivine-type silicates, M 2 SiO 4 (M – divalent cation), are a major and important component of the upper Earth’s mantle. Therefore, the properties of these materials are of considerable interest in physics, geology and crystal chemistry. Olivine compounds are used as an important composition in some refractory materials, additives in cement concrete, acid-resistant containers, ceramic pigments, etc. Well known examples of natural olivine-type silicates are fayalite (Fe 2 SiO 4 , with paramagnetic Fe 2+ ions) and forsterite (Mg 2 SiO 4 , with diamagnetic Mg 2+ ions). There are also a few natural members with another transition (Mn, Ni) or alkaline-earth (Ca) metal ions and their mixtures, e.g. tephroite (Mn 2 SiO 4 ), kirschsteinite (CaFeSiO 4 ), etc. A remarkable feature of the orthorhombic olivine-type structure (space group Pnma, no. 62 [1]) consists in two crystallographically non-equivalent M positions. Moreover, these systems are interesting due to the peculiarities of their magnetic structures. The magnetic properties of olivine compounds are quite complex and depend on type of M cation. Synthetic Co 2 SiO 4 also crystallizes in the olivine-type structure. An antiferromagnetic phase transition occurs in this compound. However, the magnetic properties of Co 2 SiO 4 were not yet well understood. Therefore, in order to determine the nature of magnetism in this system we have performed both X-ray and neutron diffraction studies as well as magnetization measurements of cobalt-olivine. A large Co 2 SiO 4 single crystal (length ~ 1.5 cm, diameter ~ 0.5 cm) was grown by the zone melting method using a mirror furnace (Inst. of Cryst., RWTH, Aachen). The phase purity was checked using the high resolution X-ray powder diffraction (MILIDI, Inst. of Cryst., RWTH, Aachen) in the temperature range from 19 to 300 K with Cu Kα radiation. The unpolarized neutron diffraction measurements were done using the single crystal diffractometer HEiDi [2] at the hot source of the FRM-II (TU Munich, Germany). Data were collected at 2 K, 55 K and 300 K with wavelength of 0.55 Å up to about sinΘ/λ ≈ 1.1 Å -1 . We have measured 3021 reflections all together with 1223 unique reflections (891 reflections with I > 3σ(I)) at 2 K. Likewise, 2232 reflections all together with 1390 unique reflections (1026 reflections with I > 3σ(I)) were collected at 55 K. At room temperature we have measured 2357 reflections with 1465 unique reflections (1092 reflections with I > 3σ(I)). Temperature stability was better than 0.1 K. The neutron diffraction data were analyzed with the Rietveld method using the FullProf program [3]. The dc magnetization measurements were performed using a Quantum Design MPMS-5 SQUID magnetometer (HMI, Berlin). The temperature dependencies of the magnetization M(T) were measured on warming from 4 to 300 K in a field of 5 T. As was already pointed out, Co 2 SiO 4 have an olivine-type orthorhombic crystal structure with the space group Pnma in which four formula units are contained in the unit cell (figure 1). The silicon atoms are coordinated with the four oxygen atoms to form SiO 4 tetrahedra. The cobalt atoms are surrounded by the six oxygen atoms and form CoO 6 octahedra. There are two crystallographically non-equivalent Co sites, where Co I (4a) ions are sites of inversion symmetry (the smaller and more distorted sites), and Co II (4c) ions are in the plane of mirror symmetry (the lager and less distorted sites). Preliminary studies of the sample at/below room temperature were carried out using X-ray diffraction, and the crystal structure is confirmed to be orthorhombic. The results indicate that the general trend of the thermal expansion appears to be normal; the unit cell parameters and the cell volume were found to gradually increase with temperature (figure 2). a (A) 10.31 10.30 10.29 10.28 b (A) 6.00 5.99 c (A) 4.785 4.780 Figure 1. A schematic representation of Co 2 SiO 4 crystal structure V (A 3 ) 4.775 296 295 294 0 50 100 150 200 250 300 T (K) Figure 2. Temperature dependency of the lattice parameters and cell volume of Co 2 SiO 4 According to experimental data, an antiferromagnetic phase transition occurs in this compound at T N ≈ 50 K (figures 3 and 4 and ref. [4]). On the other hand, no significant anomalies were observed in the cell parameters at temperatures near T N . The investigation of any subtle changes should be performed with smaller temperature steps, but this is outside the scope of the present work. 110
- Page 1 and 2:
XXIII ΠΑΝΕΛΛΗΝΙΟ ΣΥΝΕ
- Page 3 and 4:
Κοιτώντας τα πρακτ
- Page 5 and 6:
ΕΠΙΤΡΟΠΕΣ Οργανωτι
- Page 7 and 8:
ΠΡΟΓΡΑΜΜΑ ΣΥΝΕΔΡΙΟ
- Page 9 and 10:
21. Οργανικά τρανζίσ
- Page 11 and 12:
15:30 15:45 16:00 16:15 16:30 16:45
- Page 13 and 14:
41. Modeling and quantitative phase
- Page 15 and 16:
Ανοιχτή Συνεδρία «
- Page 17 and 18:
«NανοΥλικά και Νανο
- Page 19 and 20:
New materials and MOS device concep
- Page 21 and 22:
Reliability Characteristics of Rare
- Page 23 and 24:
Ο λόγος των ταχυτήτ
- Page 25 and 26:
Thus the mean R In-In is expected t
- Page 27 and 28:
FIG 1. Schematic representation of
- Page 29 and 30:
με 0.80 eV στη διεπιφά
- Page 31 and 32:
Εντοπισµός Φορέων
- Page 33 and 34:
Παρασκευή και Xαρακ
- Page 35 and 36:
Electrical Spin Injection from Fe i
- Page 37 and 38:
Electrical Spin Injection of Spin-P
- Page 39 and 40:
References [1] CH Lee, J. Meteer, V
- Page 41 and 42:
Σχήμα 1: Φωτογραφία
- Page 43 and 44:
SEM Image Layout Simulation Εικ
- Page 45 and 46:
Μελέτη Ατελειών Σε
- Page 47 and 48:
Facet-Stress-Driven Ordering in SiG
- Page 49 and 50:
νανοκρυσταλλίτης (a
- Page 51 and 52:
Σχήµα 1. Εικόνες περ
- Page 53 and 54:
Σχήµα 1. Εικόνες περ
- Page 55 and 56:
Οι δομές που αναπτύ
- Page 57 and 58:
Raman Intensity (10 -50 cm 3 ) 1,2
- Page 59 and 60:
Μελέτη της Επίδρασ
- Page 61 and 62:
Annealing Induced Dissociation of N
- Page 63 and 64:
`Εναπόθεση με Παλμι
- Page 65 and 66:
Μελέτη της Χημείας
- Page 67 and 68:
Ανάπτυξη Νέων Μεσο
- Page 69 and 70:
Application of Thermal Quadrupoles
- Page 71 and 72:
Στοχαστική προσομο
- Page 73 and 74:
Νανοτραχύτητα κατά
- Page 75 and 76:
Ευαισθησία και Δια
- Page 77 and 78: Optical Properties of CuIn 1-x Ga x
- Page 79 and 80: ανοπτημένο με λέιζ
- Page 81 and 82: Στο σχήμα 3 φαίνοντ
- Page 83 and 84: Id (mA) -0,3 -0,2 -0,1 Vg=0 Vg=-1 V
- Page 85 and 86: Strained-Si Si 1-x Ge x graded Si 1
- Page 87 and 88: Fig. 1. Laser mask movement during
- Page 89 and 90: forwarded to the back interface dur
- Page 91 and 92: Σχήμα 2: Εκθετική εξ
- Page 93 and 94: V th (V) G m,max /G m,max0 (%) I d
- Page 95 and 96: C/ C ox 1,0 0,8 0,6 0,4 0,2 0,0 -4
- Page 97 and 98: και Ta 2 O 5 , των οποίω
- Page 99 and 100: κατασκευή της. Η πα
- Page 101 and 102: ΔP (mW) 12 10 8 6 4 2 0 0 500 1000
- Page 103 and 104: υπολογίσουμε θεωρη
- Page 105 and 106: Σχήμα 2 Σύστημα ηλε
- Page 107 and 108: Μελέτη των Μηχανισ
- Page 109 and 110: Ανάπτυξη και Μελέτ
- Page 111 and 112: Structure and Magnetic Properties o
- Page 113 and 114: Δομή και Μαγνητικέ
- Page 115 and 116: Μετρήσεις Ειδικής
- Page 117 and 118: Further, almost all of the observed
- Page 119 and 120: g-factor 2.019 2.016 2.013 2.010 2.
- Page 121 and 122: ρυθμό 4 C.min -1 , έπειτ
- Page 123 and 124: ΜΕΛΕΤΗ ΤΟΥ ΦΑΙΝΟΜΕ
- Page 125 and 126: Νέοι Εξαφερίτες Ba µ
- Page 127: Crystal Structure of a new Supramol
- Page 131 and 132: Συσχέτιση πλαστική
- Page 133 and 134: Μετασχηματισμοί φά
- Page 135 and 136: Μελέτη της Επίδρασ
- Page 137 and 138: Resonant Spin Transfer Torque in Do
- Page 139 and 140: 3 η Προφορική Συνεδ
- Page 141 and 142: technology, and e-beam lithography.
- Page 143 and 144: ecause it reduces the calculation o
- Page 145 and 146: Υπολογισμός Υψηλής
- Page 147 and 148: The thermodynamic average is obtain
- Page 149 and 150: ΑΠΟΤΕΛΕΣΜΑΤΑ Στην
- Page 151 and 152: προερχόµενη είτε α
- Page 153 and 154: Combining Magnetism and Ferroelectr
- Page 155 and 156: υµένια LCMO/STO (100) πολ
- Page 157 and 158: Our scheme is illustrated in Fig. 1
- Page 159 and 160: [6] . Η μελέτη του υλι
- Page 161 and 162: τα πειραµατικά µας
- Page 163 and 164: συµπύκνωµα. Αυτό επ
- Page 165 and 166: και αναδεικνύει τρ
- Page 167 and 168: P P P P P power P copolymers P and
- Page 169 and 170: Αυτο-οργάνωση και Μ
- Page 171 and 172: Viscoelastic Response of Micelles w
- Page 173 and 174: Διηλεκτρική απόκρι
- Page 175 and 176: Light - induced Reversible Hydrophi
- Page 177 and 178: Οι παραπάνω τρεις κ
- Page 179 and 180:
AP-PH (a.u.) 0.4 0.3 0.2 0.1 0.0 0
- Page 181 and 182:
Synthesis of Polymer Brushes onto I
- Page 183 and 184:
Conformational Properties of Dendri
- Page 185 and 186:
Structure and Dynamics of Branched
- Page 187 and 188:
∆οµή και ∆υναµική
- Page 189 and 190:
Επίδραση της Τοπολ
- Page 191 and 192:
(α) (β) Σχήμα 2: (α) Απε
- Page 193 and 194:
Figure 3. Generation 4 PAMAM-H 2 O
- Page 195 and 196:
Από όλα τα παραπάνω
- Page 197 and 198:
Το PHEGMA είναι άμορφο
- Page 199 and 200:
PS HAuCl 4 P2VP Ion loading PSP2VP
- Page 201 and 202:
The nonlinear optical response of A
- Page 203 and 204:
νανοσωµατίδια. Όπω
- Page 205 and 206:
Bioactive Glass/Nanodiamonds system
- Page 207 and 208:
Energy Loss Rates of Hot Electrons
- Page 209 and 210:
Σύνθεση και Χαρακτ
- Page 211 and 212:
μπορεί να ερμηνευτ
- Page 213 and 214:
Nανοσυνθέτα Εποξει
- Page 215 and 216:
[1] S. Iijima, Nature 354, 56 (1991
- Page 217 and 218:
Figure 3: GCMC calculations for und
- Page 219 and 220:
μερών, εμφανίζοντα
- Page 221 and 222:
1.0 Reflectance 1.1 1.0 0.9 0.8 0.7
- Page 223 and 224:
στο συντελεστή διέ
- Page 225 and 226:
¿ÔÖØÑÒØÓÐØÖÐÒÒÖÒ¸
- Page 227 and 228:
Συναπόθεση Cr - Ni σε
- Page 229 and 230:
Investigation of the Structural, Mo
- Page 231 and 232:
Modification of Perlite Cementitiou
- Page 233 and 234:
Templated Sol-Gel Synthesis Of TiO
- Page 235 and 236:
Παρασκευή Υμενίων
- Page 237 and 238:
Preparation of YSZ Solid Electrolyt
- Page 239 and 240:
Σύνθεση, Ανισοτροπ
- Page 241 and 242:
Μελέτη ανθρώπινων
- Page 243 and 244:
Local Coordination of Zn and Fe in
- Page 245 and 246:
Επίδραση της Προσθ
- Page 247 and 248:
Αλκαλική Σύνθεση κ
- Page 249 and 250:
Μηχανικές Iδιότητε
- Page 251 and 252:
The Influence of Thermal Aging on t
- Page 253 and 254:
Modeling and quantitative phase ana
- Page 255 and 256:
Συμβολή στη Συντήρ
- Page 257 and 258:
Κρυσταλλική Συµπερ
- Page 259 and 260:
Σύνθεση Στερεών ∆ι
- Page 261 and 262:
Fabrication and Characterization of
- Page 263 and 264:
∆ιερεύνηση δυνατό
- Page 265 and 266:
Συγκριτική αξιολόγ
- Page 267 and 268:
6 η Προφορική Συνεδ
- Page 269 and 270:
Ηλεκτρομαγνητική Α
- Page 271 and 272:
Φασματοσκοπική Μελ
- Page 273 and 274:
Thermal and Electrical Properties o
- Page 275 and 276:
Structure, Mechanical, and Optoelec
- Page 277 and 278:
Designing Nanoporous Materials for
- Page 279 and 280:
This program was developed to serve
- Page 281 and 282:
Νέα Αυτό-οργανούμε
- Page 283 and 284:
A Physical Model to Interpret the E
- Page 285 and 286:
FIR study of Ag x (As 33 S 33 Se 33
- Page 287 and 288:
Mελέτη Μεικτών Γυαλ
- Page 289 and 290:
The Structural Role of Fe and Zn in
- Page 291 and 292:
ΕΥΡΕΤΗΡΙΟ ΣΥΓΓΡΑΦΕ
- Page 293 and 294:
Κομπίτσας Μ…………
- Page 295 and 296:
ΕΥΡΕΤΗΡΙΟ ΣΥΓΓΡΑΦΕ
- Page 297:
W Watson I.M………………4 Weg