Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
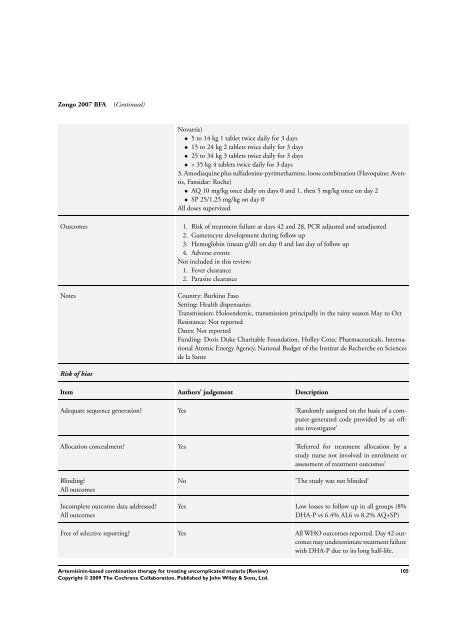
Zongo 2007 BFA (Continued)<br />
Novartis)<br />
• 5 to 14 kg 1 tablet twice daily <strong>for</strong> 3 days<br />
• 15 to 24 kg 2 tablets twice daily <strong>for</strong> 3 days<br />
• 25 to 34 kg 3 tablets twice daily <strong>for</strong> 3 days<br />
• > 35 kg 4 tablets twice daily <strong>for</strong> 3 days<br />
3. Amodiaquine plus sulfadoxine-pyrimethamine, loose <strong>combination</strong> (Flavoquine: Aventis,<br />
Fansidar: Roche)<br />
• AQ 10 mg/kg once daily on days 0 and 1, then 5 mg/kg once on day 2<br />
• SP 25/1.25 mg/kg on day 0<br />
All doses supervized<br />
Outcomes 1. Risk of treatment failure at days 42 and 28, PCR adjusted and unadjusted<br />
2. Gametocyte development during follow up<br />
3. Hemoglobin (mean g/dl) on day 0 and last day of follow up<br />
4. Adverse events<br />
Not included in this review:<br />
1. Fever clearance<br />
2. Parasite clearance<br />
Notes Country: Burkino Faso<br />
Setting: Health dispensaries<br />
Transmission: Holoendemic, transmission principally in the rainy season May to Oct<br />
Resistance: Not reported<br />
Dates: Not reported<br />
Funding: Doris Duke Charitable Foundation, Holley Cotec Pharmaceuticals, International<br />
Atomic Energy Agency, National Budget of the Institut de Recherche en Sciences<br />
de la Sante<br />
Risk of bias<br />
Item Authors’ judgement Description<br />
Adequate sequence generation? Yes ’Randomly assigned on the basis of a computer-generated<br />
code provided by an offsite<br />
investigator’<br />
Allocation concealment? Yes ’Referred <strong>for</strong> treatment allocation by a<br />
study nurse not involved in enrolment or<br />
assessment of treatment outcomes’<br />
Blinding?<br />
All outcomes<br />
Incomplete outcome data addressed?<br />
All outcomes<br />
No ’<strong>The</strong> study was not blinded’<br />
Yes Low losses to follow up in all groups (8%<br />
DHA-P vs 6.4% AL6 vs 8.2% AQ+SP)<br />
Free of selective reporting? Yes All WHO outcomes reported. Day 42 outcomes<br />
may underestimate treatment failure<br />
with DHA-P due to its long half-life.<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
105