Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
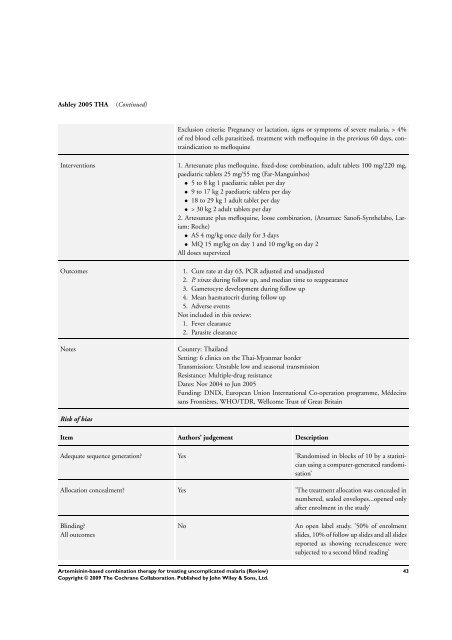
Ashley 2005 THA (Continued)<br />
Exclusion criteria: Pregnancy or lactation, signs or symptoms of severe malaria, > 4%<br />
of red blood cells parasitized, treatment with mefloquine in the previous 60 days, contraindication<br />
to mefloquine<br />
Interventions 1. Artesunate plus mefloquine, fixed-dose <strong>combination</strong>, adult tablets 100 mg/220 mg,<br />
paediatric tablets 25 mg/55 mg (Far-Manguinhos)<br />
• 5 to 8 kg 1 paediatric tablet per day<br />
• 9 to 17 kg 2 paediatric tablets per day<br />
• 18 to 29 kg 1 adult tablet per day<br />
• > 30 kg 2 adult tablets per day<br />
2. Artesunate plus mefloquine, loose <strong>combination</strong>, (Arsumax: Sanofi-Synthelabo, Lariam:<br />
Roche)<br />
• AS 4 mg/kg once daily <strong>for</strong> 3 days<br />
• MQ 15 mg/kg on day 1 and 10 mg/kg on day 2<br />
All doses supervized<br />
Outcomes 1. Cure rate at day 63, PCR adjusted and unadjusted<br />
2. P. vivax during follow up, and median time to reappearance<br />
3. Gametocyte development during follow up<br />
4. Mean haematocrit during follow up<br />
5. Adverse events<br />
Not included in this review:<br />
1. Fever clearance<br />
2. Parasite clearance<br />
Notes Country: Thailand<br />
Setting: 6 clinics on the Thai-Myanmar border<br />
Transmission: Unstable low and seasonal transmission<br />
Resistance: Multiple-drug resistance<br />
Dates: Nov 2004 to Jun 2005<br />
Funding: DNDi, European Union International Co-operation programme, Médecins<br />
sans Frontières, WHO/TDR, Wellcome Trust of Great Britain<br />
Risk of bias<br />
Item Authors’ judgement Description<br />
Adequate sequence generation? Yes ’Randomised in blocks of 10 by a statistician<br />
using a computer-generated randomisation’<br />
Allocation concealment? Yes ’<strong>The</strong> treatment allocation was concealed in<br />
numbered, sealed envelopes...opened only<br />
after enrolment in the study’<br />
Blinding?<br />
All outcomes<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
No An open label study. ’50% of enrolment<br />
slides, 10% of follow up slides and all slides<br />
reported as showing recrudescence were<br />
subjected to a second blind reading’<br />
43