Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
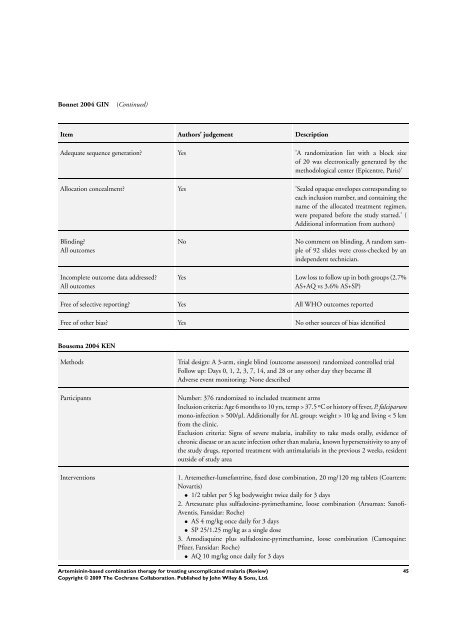
Bonnet 2004 GIN (Continued)<br />
Item Authors’ judgement Description<br />
Adequate sequence generation? Yes ’A randomization list with a block size<br />
of 20 was electronically generated by the<br />
methodological center (Epicentre, Paris)’<br />
Allocation concealment? Yes ’Sealed opaque envelopes corresponding to<br />
each inclusion number, and containing the<br />
name of the allocated treatment regimen,<br />
were prepared be<strong>for</strong>e the study started.’ (<br />
Additional in<strong>for</strong>mation from authors)<br />
Blinding?<br />
All outcomes<br />
Incomplete outcome data addressed?<br />
All outcomes<br />
No No comment on blinding. A random sample<br />
of 92 slides were cross-checked by an<br />
independent technician.<br />
Yes Low loss to follow up in both groups (2.7%<br />
AS+AQ vs 3.6% AS+SP)<br />
Free of selective reporting? Yes All WHO outcomes reported<br />
Free of other bias? Yes No other sources of bias identified<br />
Bousema 2004 KEN<br />
Methods Trial design: A 3-arm, single blind (outcome assessors) randomized controlled trial<br />
Follow up: Days 0, 1, 2, 3, 7, 14, and 28 or any other day they became ill<br />
Adverse event monitoring: None described<br />
Participants Number: 376 randomized to included treatment arms<br />
Inclusion criteria: Age 6 months to 10 yrs, temp > 37.5 ºC or history of fever, P. falciparum<br />
mono-infection > 500/µl. Additionally <strong>for</strong> AL group: weight > 10 kg and living < 5 km<br />
from the clinic.<br />
Exclusion criteria: Signs of severe malaria, inability to take meds orally, evidence of<br />
chronic disease or an acute infection other than malaria, known hypersensitivity to any of<br />
the study drugs, reported treatment with antimalarials in the previous 2 weeks, resident<br />
outside of study area<br />
Interventions 1. Artemether-lumefantrine, fixed dose <strong>combination</strong>, 20 mg/120 mg tablets (Coartem:<br />
Novartis)<br />
• 1/2 tablet per 5 kg bodyweight twice daily <strong>for</strong> 3 days<br />
2. Artesunate plus sulfadoxine-pyrimethamine, loose <strong>combination</strong> (Arsumax: Sanofi-<br />
Aventis, Fansidar: Roche)<br />
• AS 4 mg/kg once daily <strong>for</strong> 3 days<br />
• SP 25/1.25 mg/kg as a single dose<br />
3. Amodiaquine plus sulfadoxine-pyrimethamine, loose <strong>combination</strong> (Camoquine:<br />
Pfizer, Fansidar: Roche)<br />
• AQ 10 mg/kg once daily <strong>for</strong> 3 days<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
45