Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
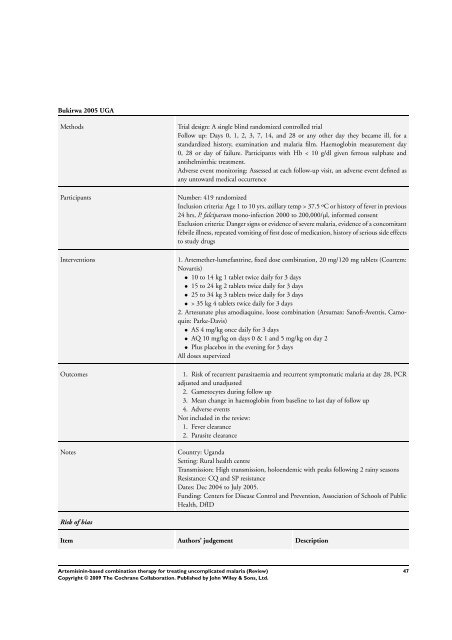
Bukirwa 2005 UGA<br />
Methods Trial design: A single blind randomized controlled trial<br />
Follow up: Days 0, 1, 2, 3, 7, 14, and 28 or any other day they became ill, <strong>for</strong> a<br />
standardized history, examination and malaria film. Haemoglobin measurement day<br />
0, 28 or day of failure. Participants with Hb < 10 g/dl given ferrous sulphate and<br />
antihelminthic treatment.<br />
Adverse event monitoring: Assessed at each follow-up visit, an adverse event defined as<br />
any untoward medical occurrence<br />
Participants Number: 419 randomized<br />
Inclusion criteria: Age 1 to 10 yrs, axillary temp > 37.5 ºC or history of fever in previous<br />
24 hrs, P. falciparum mono-infection 2000 to 200,000/µl, in<strong>for</strong>med consent<br />
Exclusion criteria: Danger signs or evidence of severe malaria, evidence of a concomitant<br />
febrile illness, repeated vomiting of first dose of medication, history of serious side effects<br />
to study drugs<br />
Interventions 1. Artemether-lumefantrine, fixed dose <strong>combination</strong>, 20 mg/120 mg tablets (Coartem:<br />
Novartis)<br />
• 10 to 14 kg 1 tablet twice daily <strong>for</strong> 3 days<br />
• 15 to 24 kg 2 tablets twice daily <strong>for</strong> 3 days<br />
• 25 to 34 kg 3 tablets twice daily <strong>for</strong> 3 days<br />
• > 35 kg 4 tablets twice daily <strong>for</strong> 3 days<br />
2. Artesunate plus amodiaquine, loose <strong>combination</strong> (Arsumax: Sanofi-Aventis, Camoquin:<br />
Parke-Davis)<br />
• AS 4 mg/kg once daily <strong>for</strong> 3 days<br />
• AQ 10 mg/kg on days 0 & 1 and 5 mg/kg on day 2<br />
• Plus placebos in the evening <strong>for</strong> 3 days<br />
All doses supervized<br />
Outcomes 1. Risk of recurrent parasitaemia and recurrent symptomatic malaria at day 28, PCR<br />
adjusted and unadjusted<br />
2. Gametocytes during follow up<br />
3. Mean change in haemoglobin from baseline to last day of follow up<br />
4. Adverse events<br />
Not included in the review:<br />
1. Fever clearance<br />
2. Parasite clearance<br />
Notes Country: Uganda<br />
Setting: Rural health centre<br />
Transmission: High transmission, holoendemic with peaks following 2 rainy seasons<br />
Resistance: CQ and SP resistance<br />
Dates: Dec 2004 to July 2005.<br />
Funding: Centers <strong>for</strong> Disease Control and Prevention, Association of Schools of Public<br />
Health, DfID<br />
Risk of bias<br />
Item Authors’ judgement Description<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
47