Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
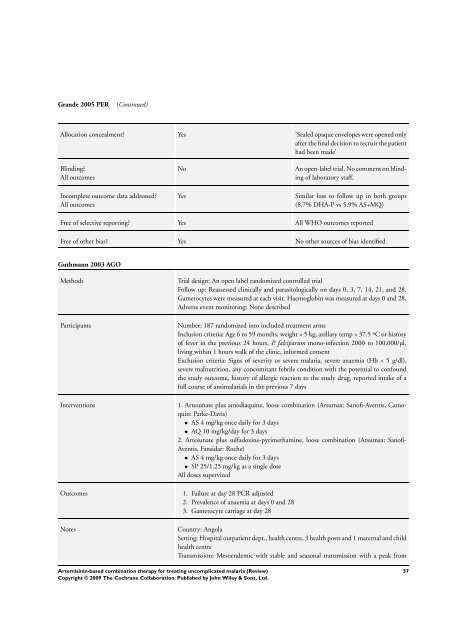
Grande 2005 PER (Continued)<br />
Allocation concealment? Yes ’Sealed opaque envelopes were opened only<br />
after the final decision to recruit the patient<br />
had been made’<br />
Blinding?<br />
All outcomes<br />
Incomplete outcome data addressed?<br />
All outcomes<br />
No An open-label trial. No comment on blinding<br />
of laboratory staff.<br />
Yes Similar loss to follow up in both groups<br />
(8.7% DHA-P vs 5.9% AS+MQ)<br />
Free of selective reporting? Yes All WHO outcomes reported<br />
Free of other bias? Yes No other sources of bias identified<br />
Guthmann 2003 AGO<br />
Methods Trial design: An open label randomized controlled trial<br />
Follow up: Reassessed clinically and parasitologically on days 0, 3, 7, 14, 21, and 28.<br />
Gametocytes were measured at each visit. Haemoglobin was measured at days 0 and 28.<br />
Adverse event monitoring: None described<br />
Participants Number: 187 randomized into included treatment arms<br />
Inclusion criteria: Age 6 to 59 months, weight > 5 kg, axillary temp > 37.5 ºC or history<br />
of fever in the previous 24 hours, P. falciparum mono-infection 2000 to 100,000/µl,<br />
living within 1 hours walk of the clinic, in<strong>for</strong>med consent<br />
Exclusion criteria: Signs of severity or severe malaria, severe anaemia (Hb < 5 g/dl),<br />
severe malnutrition, any concomitant febrile condition with the potential to confound<br />
the study outcome, history of allergic reaction to the study drug, reported intake of a<br />
full course of antimalarials in the previous 7 days<br />
Interventions 1. Artesunate plus amodiaquine, loose <strong>combination</strong> (Arsumax: Sanofi-Aventis, Camoquin:<br />
Parke-Davis)<br />
• AS 4 mg/kg once daily <strong>for</strong> 3 days<br />
• AQ 10 mg/kg/day <strong>for</strong> 3 days<br />
2. Artesunate plus sulfadoxine-pyrimethamine, loose <strong>combination</strong> (Arsumax: Sanofi-<br />
Aventis, Fansidar: Roche)<br />
• AS 4 mg/kg once daily <strong>for</strong> 3 days<br />
• SP 25/1.25 mg/kg as a single dose<br />
All doses supervized<br />
Outcomes 1. Failure at day 28 PCR adjusted<br />
2. Prevalence of anaemia at days 0 and 28<br />
3. Gametocyte carriage at day 28<br />
Notes Country: Angola<br />
Setting: Hospital outpatient dept., health centre, 3 health posts and 1 maternal and child<br />
health centre<br />
Transmission: Mesoendemic with stable and seasonal transmission with a peak from<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
57