Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
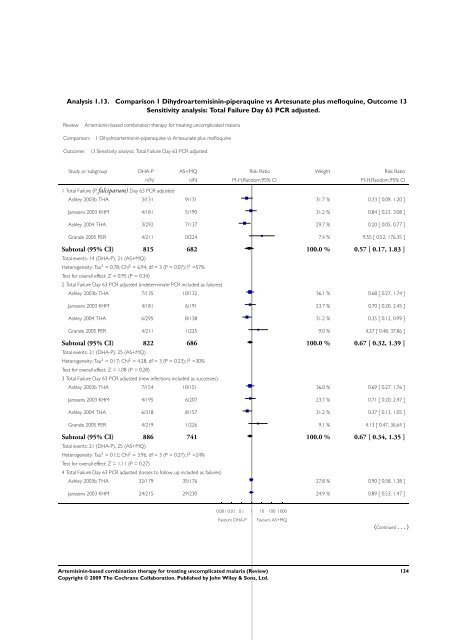
Analysis 1.13. Comparison 1 Dihydroartemisinin-piperaquine vs Artesunate plus mefloquine, Outcome 13<br />
Sensitivity analysis: Total Failure Day 63 PCR adjusted.<br />
Review: <strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria<br />
Comparison: 1 Dihydroartemisinin-piperaquine vs Artesunate plus mefloquine<br />
Outcome: 13 Sensitivity analysis: Total Failure Day 63 PCR adjusted<br />
Study or subgroup DHA-P AS+MQ Risk Ratio Weight Risk Ratio<br />
1 Total Failure (P. falciparum) Day 63 PCR adjusted<br />
n/N n/N M-H,Random,95% CI M-H,Random,95% CI<br />
Ashley 2003b THA 3/131 9/131 31.7 % 0.33 [ 0.09, 1.20 ]<br />
Janssens 2003 KHM 4/181 5/190 31.2 % 0.84 [ 0.23, 3.08 ]<br />
Ashley 2004 THA 3/292 7/137 29.7 % 0.20 [ 0.05, 0.77 ]<br />
Grande 2005 PER 4/211 0/224 7.4 % 9.55 [ 0.52, 176.35 ]<br />
Subtotal (95% CI) 815 682 100.0 % 0.57 [ 0.17, 1.83 ]<br />
Total events: 14 (DHA-P), 21 (AS+MQ)<br />
Heterogeneity: Tau 2 = 0.78; Chi 2 = 6.94, df = 3 (P = 0.07); I 2 =57%<br />
Test <strong>for</strong> overall effect: Z = 0.95 (P = 0.34)<br />
2 Total Failure Day 63 PCR adjusted (indeterminate PCR included as failures)<br />
Ashley 2003b THA 7/135 10/132 36.1 % 0.68 [ 0.27, 1.74 ]<br />
Janssens 2003 KHM 4/181 6/191 23.7 % 0.70 [ 0.20, 2.45 ]<br />
Ashley 2004 THA 6/295 8/138 31.2 % 0.35 [ 0.12, 0.99 ]<br />
Grande 2005 PER 4/211 1/225 9.0 % 4.27 [ 0.48, 37.86 ]<br />
Subtotal (95% CI) 822 686 100.0 % 0.67 [ 0.32, 1.39 ]<br />
Total events: 21 (DHA-P), 25 (AS+MQ)<br />
Heterogeneity: Tau 2 = 0.17; Chi 2 = 4.28, df = 3 (P = 0.23); I 2 =30%<br />
Test <strong>for</strong> overall effect: Z = 1.08 (P = 0.28)<br />
3 Total Failure Day 63 PCR adjusted (new infections included as successes)<br />
Ashley 2003b THA 7/154 10/151 36.0 % 0.69 [ 0.27, 1.76 ]<br />
Janssens 2003 KHM 4/195 6/207 23.7 % 0.71 [ 0.20, 2.47 ]<br />
Ashley 2004 THA 6/318 8/157 31.2 % 0.37 [ 0.13, 1.05 ]<br />
Grande 2005 PER 4/219 1/226 9.1 % 4.13 [ 0.47, 36.64 ]<br />
Subtotal (95% CI) 886 741 100.0 % 0.67 [ 0.34, 1.35 ]<br />
Total events: 21 (DHA-P), 25 (AS+MQ)<br />
Heterogeneity: Tau 2 = 0.12; Chi 2 = 3.96, df = 3 (P = 0.27); I 2 =24%<br />
Test <strong>for</strong> overall effect: Z = 1.11 (P = 0.27)<br />
4 Total Failure Day 63 PCR adjusted (losses to follow up included as failures)<br />
Ashley 2003b THA 32/179 35/176 27.8 % 0.90 [ 0.58, 1.38 ]<br />
Janssens 2003 KHM 24/215 29/230 24.9 % 0.89 [ 0.53, 1.47 ]<br />
0.001 0.01 0.1 1 10 100 1000<br />
Favours DHA-P Favours AS+MQ<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
(Continued ...)<br />
134