Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
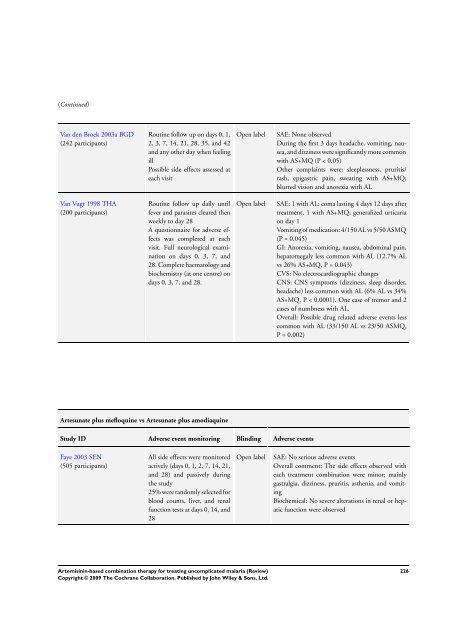
(Continued)<br />
Van den Broek 2003a BGD<br />
(242 participants)<br />
Van Vugt 1998 THA<br />
(200 participants)<br />
Routine follow up on days 0, 1,<br />
2, 3, 7, 14, 21, 28, 35, and 42<br />
and any other day when feeling<br />
ill<br />
Possible side effects assessed at<br />
each visit<br />
Routine follow up daily until<br />
fever and parasites cleared then<br />
weekly to day 28<br />
A questionnaire <strong>for</strong> adverse effects<br />
was completed at each<br />
visit. Full neurological examination<br />
on days 0, 3, 7, and<br />
28. Complete haematology and<br />
biochemistry (at one centre) on<br />
days 0, 3, 7, and 28.<br />
Artesunate plus mefloquine vs Artesunate plus amodiaquine<br />
Study ID Adverse event monitoring Blinding Adverse events<br />
Faye 2003 SEN<br />
(505 participants)<br />
All side effects were monitored<br />
actively (days 0, 1, 2, 7, 14, 21,<br />
and 28) and passively during<br />
the study<br />
25% were randomly selected <strong>for</strong><br />
blood counts, liver, and renal<br />
function tests at days 0, 14, and<br />
28<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
Open label SAE: None observed<br />
During the first 3 days headache, vomiting, nausea,<br />
and dizziness were significantly more common<br />
with AS+MQ (P < 0.05)<br />
Other complaints were: sleeplessness, pruritis/<br />
rash, epigastric pain, sweating with AS+MQ;<br />
blurred vision and anorexia with AL<br />
Open label SAE: 1 with AL: coma lasting 4 days 12 days after<br />
treatment, 1 with AS+MQ; generalized urticaria<br />
on day 1<br />
Vomiting of medication: 4/150 AL vs 5/50 ASMQ<br />
(P = 0.045)<br />
GI: Anorexia, vomiting, nausea, abdominal pain,<br />
hepatomegaly less common with AL (12.7% AL<br />
vs 26% AS+MQ, P = 0.043)<br />
CVS: No electrocardiographic changes<br />
CNS: CNS symptoms (dizziness, sleep disorder,<br />
headache) less common with AL (6% AL vs 34%<br />
AS+MQ, P < 0.0001). One case of tremor and 2<br />
cases of numbness with AL.<br />
Overall: Possible drug related adverse events less<br />
common with AL (33/150 AL vs 23/50 ASMQ,<br />
P = 0.002)<br />
Open label SAE: No serious adverse events<br />
Overall comment: <strong>The</strong> side effects observed with<br />
each treatment <strong>combination</strong> were minor; mainly<br />
gastralgia, dizziness, pruritis, asthenia, and vomiting<br />
Biochemical: No severe alterations in renal or hepatic<br />
function were observed<br />
226