Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
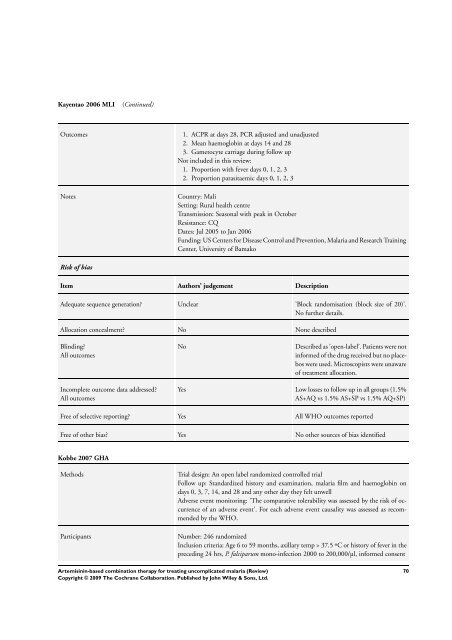
Kayentao 2006 MLI (Continued)<br />
Outcomes 1. ACPR at days 28, PCR adjusted and unadjusted<br />
2. Mean haemoglobin at days 14 and 28<br />
3. Gametocyte carriage during follow up<br />
Not included in this review:<br />
1. Proportion with fever days 0, 1, 2, 3<br />
2. Proportion parasitaemic days 0, 1, 2, 3<br />
Notes Country: Mali<br />
Setting: Rural health centre<br />
Transmission: Seasonal with peak in October<br />
Resistance: CQ<br />
Dates: Jul 2005 to Jan 2006<br />
Funding: US Centers <strong>for</strong> Disease Control and Prevention, Malaria and Research Training<br />
Center, University of Bamako<br />
Risk of bias<br />
Item Authors’ judgement Description<br />
Adequate sequence generation? Unclear ’Block randomisation (block size of 20)’.<br />
No further details.<br />
Allocation concealment? No None described<br />
Blinding?<br />
All outcomes<br />
Incomplete outcome data addressed?<br />
All outcomes<br />
No Described as ’open-label’. Patients were not<br />
in<strong>for</strong>med of the drug received but no placebos<br />
were used. Microscopists were unaware<br />
of treatment allocation.<br />
Yes Low losses to follow up in all groups (1.5%<br />
AS+AQ vs 1.5% AS+SP vs 1.5% AQ+SP)<br />
Free of selective reporting? Yes All WHO outcomes reported<br />
Free of other bias? Yes No other sources of bias identified<br />
Kobbe 2007 GHA<br />
Methods Trial design: An open label randomized controlled trial<br />
Follow up: Standardized history and examination, malaria film and haemoglobin on<br />
days 0, 3, 7, 14, and 28 and any other day they felt unwell<br />
Adverse event monitoring: ’<strong>The</strong> comparative tolerability was assessed by the risk of occurrence<br />
of an adverse event’. For each adverse event causality was assessed as recommended<br />
by the WHO.<br />
Participants Number: 246 randomized<br />
Inclusion criteria: Age 6 to 59 months, axillary temp > 37.5 ºC or history of fever in the<br />
preceding 24 hrs, P. falciparum mono-infection 2000 to 200,000/µl, in<strong>for</strong>med consent<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
70