Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
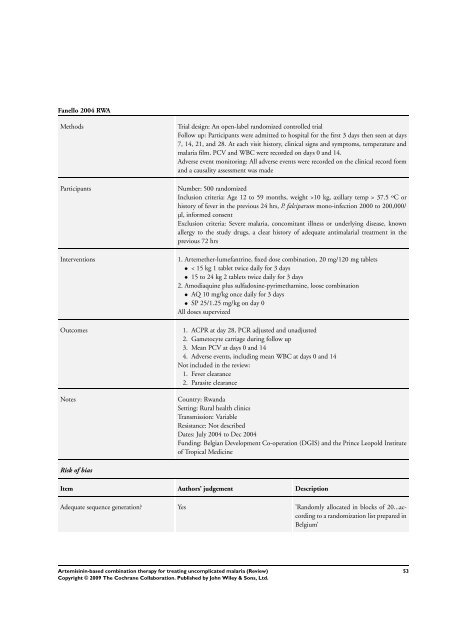
Fanello 2004 RWA<br />
Methods Trial design: An open-label randomized controlled trial<br />
Follow up: Participants were admitted to hospital <strong>for</strong> the first 3 days then seen at days<br />
7, 14, 21, and 28. At each visit history, clinical signs and symptoms, temperature and<br />
malaria film. PCV and WBC were recorded on days 0 and 14.<br />
Adverse event monitoring: All adverse events were recorded on the clinical record <strong>for</strong>m<br />
and a causality assessment was made<br />
Participants Number: 500 randomized<br />
Inclusion criteria: Age 12 to 59 months, weight >10 kg, axillary temp > 37.5 ºC or<br />
history of fever in the previous 24 hrs, P. falciparum mono-infection 2000 to 200,000/<br />
µl, in<strong>for</strong>med consent<br />
Exclusion criteria: Severe malaria, concomitant illness or underlying disease, known<br />
allergy to the study drugs, a clear history of adequate antimalarial treatment in the<br />
previous 72 hrs<br />
Interventions 1. Artemether-lumefantrine, fixed dose <strong>combination</strong>, 20 mg/120 mg tablets<br />
• < 15 kg 1 tablet twice daily <strong>for</strong> 3 days<br />
• 15 to 24 kg 2 tablets twice daily <strong>for</strong> 3 days<br />
2. Amodiaquine plus sulfadoxine-pyrimethamine, loose <strong>combination</strong><br />
• AQ 10 mg/kg once daily <strong>for</strong> 3 days<br />
• SP 25/1.25 mg/kg on day 0<br />
All doses supervized<br />
Outcomes 1. ACPR at day 28, PCR adjusted and unadjusted<br />
2. Gametocyte carriage during follow up<br />
3. Mean PCV at days 0 and 14<br />
4. Adverse events, including mean WBC at days 0 and 14<br />
Not included in the review:<br />
1. Fever clearance<br />
2. Parasite clearance<br />
Notes Country: Rwanda<br />
Setting: Rural health clinics<br />
Transmission: Variable<br />
Resistance: Not described<br />
Dates: July 2004 to Dec 2004<br />
Funding: Belgian Development Co-operation (DGIS) and the Prince Leopold Institute<br />
of Tropical Medicine<br />
Risk of bias<br />
Item Authors’ judgement Description<br />
Adequate sequence generation? Yes ’Randomly allocated in blocks of 20...according<br />
to a randomization list prepared in<br />
Belgium’<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
53