Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
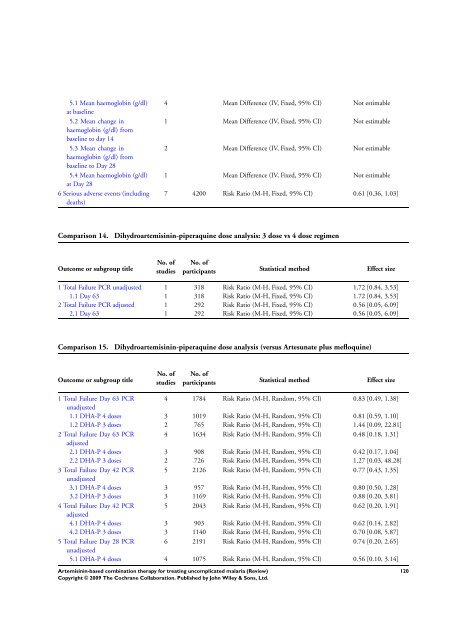
5.1 Mean haemoglobin (g/dl)<br />
at baseline<br />
5.2 Mean change in<br />
haemoglobin (g/dl) from<br />
baseline to day 14<br />
5.3 Mean change in<br />
haemoglobin (g/dl) from<br />
baseline to Day 28<br />
5.4 Mean haemoglobin (g/dl)<br />
at Day 28<br />
6 Serious adverse events (including<br />
deaths)<br />
4 Mean Difference (IV, Fixed, 95% CI) Not estimable<br />
1 Mean Difference (IV, Fixed, 95% CI) Not estimable<br />
2 Mean Difference (IV, Fixed, 95% CI) Not estimable<br />
1 Mean Difference (IV, Fixed, 95% CI) Not estimable<br />
7 4200 Risk Ratio (M-H, Fixed, 95% CI) 0.61 [0.36, 1.03]<br />
Comparison 14. Dihydroartemisinin-piperaquine dose analysis: 3 dose vs 4 dose regimen<br />
Outcome or subgroup title<br />
No. of<br />
studies<br />
No. of<br />
participants Statistical method Effect size<br />
1 Total Failure PCR unadjusted 1 318 Risk Ratio (M-H, Fixed, 95% CI) 1.72 [0.84, 3.53]<br />
1.1 Day 63 1 318 Risk Ratio (M-H, Fixed, 95% CI) 1.72 [0.84, 3.53]<br />
2 Total Failure PCR adjusted 1 292 Risk Ratio (M-H, Fixed, 95% CI) 0.56 [0.05, 6.09]<br />
2.1 Day 63 1 292 Risk Ratio (M-H, Fixed, 95% CI) 0.56 [0.05, 6.09]<br />
Comparison 15. Dihydroartemisinin-piperaquine dose analysis (versus Artesunate plus mefloquine)<br />
Outcome or subgroup title<br />
No. of<br />
studies<br />
No. of<br />
participants Statistical method Effect size<br />
1 Total Failure Day 63 PCR<br />
unadjusted<br />
4 1784 Risk Ratio (M-H, Random, 95% CI) 0.83 [0.49, 1.38]<br />
1.1 DHA-P 4 doses 3 1019 Risk Ratio (M-H, Random, 95% CI) 0.81 [0.59, 1.10]<br />
1.2 DHA-P 3 doses 2 765 Risk Ratio (M-H, Random, 95% CI) 1.44 [0.09, 22.81]<br />
2 Total Failure Day 63 PCR<br />
adjusted<br />
4 1634 Risk Ratio (M-H, Random, 95% CI) 0.48 [0.18, 1.31]<br />
2.1 DHA-P 4 doses 3 908 Risk Ratio (M-H, Random, 95% CI) 0.42 [0.17, 1.04]<br />
2.2 DHA-P 3 doses 2 726 Risk Ratio (M-H, Random, 95% CI) 1.27 [0.03, 48.28]<br />
3 Total Failure Day 42 PCR<br />
unadjusted<br />
5 2126 Risk Ratio (M-H, Random, 95% CI) 0.77 [0.43, 1.35]<br />
3.1 DHA-P 4 doses 3 957 Risk Ratio (M-H, Random, 95% CI) 0.80 [0.50, 1.28]<br />
3.2 DHA-P 3 doses 3 1169 Risk Ratio (M-H, Random, 95% CI) 0.88 [0.20, 3.81]<br />
4 Total Failure Day 42 PCR<br />
adjusted<br />
5 2043 Risk Ratio (M-H, Random, 95% CI) 0.62 [0.20, 1.91]<br />
4.1 DHA-P 4 doses 3 903 Risk Ratio (M-H, Random, 95% CI) 0.62 [0.14, 2.82]<br />
4.2 DHA-P 3 doses 3 1140 Risk Ratio (M-H, Random, 95% CI) 0.70 [0.08, 5.87]<br />
5 Total Failure Day 28 PCR<br />
unadjusted<br />
6 2191 Risk Ratio (M-H, Random, 95% CI) 0.74 [0.20, 2.65]<br />
5.1 DHA-P 4 doses 4 1075 Risk Ratio (M-H, Random, 95% CI) 0.56 [0.10, 3.14]<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
120