Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
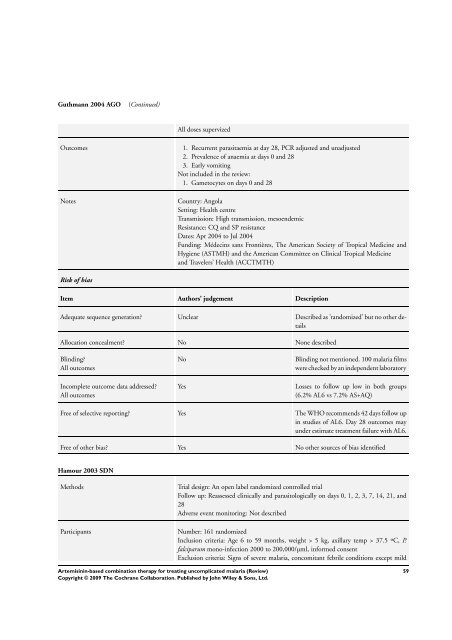
Guthmann 2004 AGO (Continued)<br />
All doses supervized<br />
Outcomes 1. Recurrent parasitaemia at day 28, PCR adjusted and unadjusted<br />
2. Prevalence of anaemia at days 0 and 28<br />
3. Early vomiting<br />
Not included in the review:<br />
1. Gametocytes on days 0 and 28<br />
Notes Country: Angola<br />
Setting: Health centre<br />
Transmission: High transmission, mesoendemic<br />
Resistance: CQ and SP resistance<br />
Dates: Apr 2004 to Jul 2004<br />
Funding: Médecins sans Frontières, <strong>The</strong> American Society of Tropical Medicine and<br />
Hygiene (ASTMH) and the American Committee on Clinical Tropical Medicine<br />
and Travelers’ Health (ACCTMTH)<br />
Risk of bias<br />
Item Authors’ judgement Description<br />
Adequate sequence generation? Unclear Described as ’randomized’ but no other details<br />
Allocation concealment? No None described<br />
Blinding?<br />
All outcomes<br />
Incomplete outcome data addressed?<br />
All outcomes<br />
No Blinding not mentioned. 100 malaria films<br />
were checked by an independent laboratory<br />
Yes Losses to follow up low in both groups<br />
(6.2% AL6 vs 7.2% AS+AQ)<br />
Free of selective reporting? Yes <strong>The</strong> WHO recommends 42 days follow up<br />
in studies of AL6. Day 28 outcomes may<br />
under estimate treatment failure with AL6.<br />
Free of other bias? Yes No other sources of bias identified<br />
Hamour 2003 SDN<br />
Methods Trial design: An open label randomized controlled trial<br />
Follow up: Reassessed clinically and parasitologically on days 0, 1, 2, 3, 7, 14, 21, and<br />
28<br />
Adverse event monitoring: Not described<br />
Participants Number: 161 randomized<br />
Inclusion criteria: Age 6 to 59 months, weight > 5 kg, axillary temp > 37.5 ºC, P.<br />
falciparum mono-infection 2000 to 200,000/µml, in<strong>for</strong>med consent<br />
Exclusion criteria: Signs of severe malaria, concomitant febrile conditions except mild<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
59