Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
(Continued)<br />
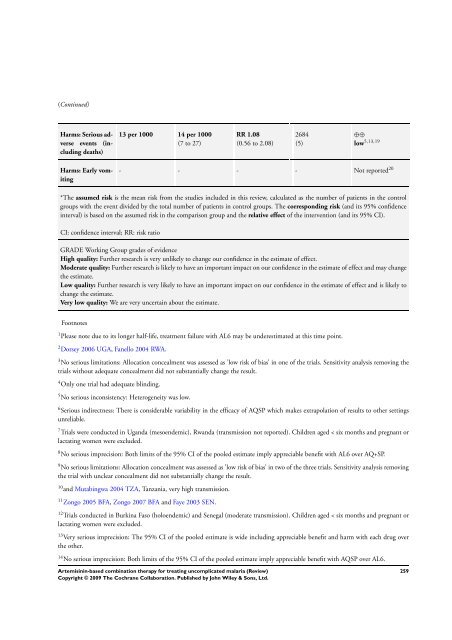
Harms: Serious adverse<br />
events (including<br />
deaths)<br />
Harms: Early vomiting<br />
13 per 1000 14 per 1000<br />
(7 to 27)<br />
RR 1.08<br />
(0.56 to 2.08)<br />
2684<br />
(5)<br />
⊕⊕<br />
low 5,13,19<br />
- - - - Not reported 20<br />
*<strong>The</strong> assumed risk is the mean risk from the studies included in this review, calculated as the number of patients in the control<br />
groups with the event divided by the total number of patients in control groups. <strong>The</strong> corresponding risk (and its 95% confidence<br />
interval) is <strong>based</strong> on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).<br />
CI: confidence interval; RR: risk ratio<br />
GRADE Working Group grades of evidence<br />
High quality: Further research is very unlikely to change our confidence in the estimate of effect.<br />
Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change<br />
the estimate.<br />
Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to<br />
change the estimate.<br />
Very low quality: We are very uncertain about the estimate.<br />
Footnotes<br />
1 Please note due to its longer half-life, treatment failure with AL6 may be underestimated at this time point.<br />
2 Dorsey 2006 UGA, Fanello 2004 RWA.<br />
3 No serious limitations: Allocation concealment was assessed as ’low risk of bias’ in one of the trials. Sensitivity analysis removing the<br />
trials without adequate concealment did not substantially change the result.<br />
4 Only one trial had adequate blinding.<br />
5 No serious inconsistency: Heterogeneity was low.<br />
6 Serious indirectness: <strong>The</strong>re is considerable variability in the efficacy of AQSP which makes extrapolation of results to other settings<br />
unreliable.<br />
7 Trials were conducted in Uganda (mesoendemic), Rwanda (transmission not reported). Children aged < six months and pregnant or<br />
lactating women were excluded.<br />
8 No serious imprecision: Both limits of the 95% CI of the pooled estimate imply appreciable benefit with AL6 over AQ+SP.<br />
9 No serious limitations: Allocation concealment was assessed as ’low risk of bias’ in two of the three trials. Sensitivity analysis removing<br />
the trial with unclear concealment did not substantially change the result.<br />
10 and Mutabingwa 2004 TZA, Tanzania, very high transmission.<br />
11 Zongo 2005 BFA, Zongo 2007 BFA and Faye 2003 SEN.<br />
12 Trials conducted in Burkina Faso (holoendemic) and Senegal (moderate transmission). Children aged < six months and pregnant or<br />
lactating women were excluded.<br />
13 Very serious imprecision: <strong>The</strong> 95% CI of the pooled estimate is wide including appreciable benefit and harm with each drug over<br />
the other.<br />
14 No serious imprecision: Both limits of the 95% CI of the pooled estimate imply appreciable benefit with AQSP over AL6.<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
259