Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
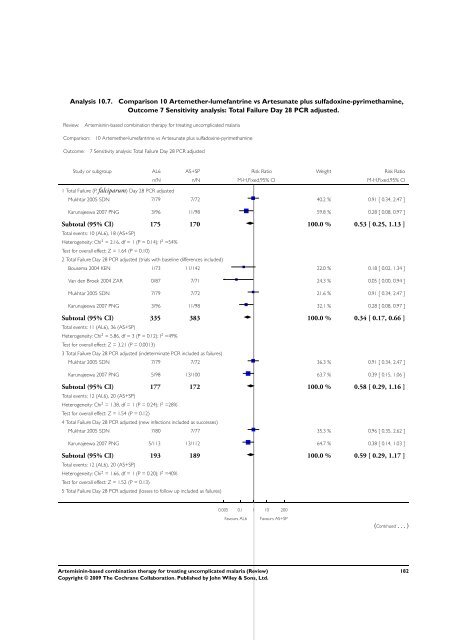
Analysis 10.7. Comparison 10 Artemether-lumefantrine vs Artesunate plus sulfadoxine-pyrimethamine,<br />
Outcome 7 Sensitivity analysis: Total Failure Day 28 PCR adjusted.<br />
Review: <strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria<br />
Comparison: 10 Artemether-lumefantrine vs Artesunate plus sulfadoxine-pyrimethamine<br />
Outcome: 7 Sensitivity analysis: Total Failure Day 28 PCR adjusted<br />
Study or subgroup AL6 AS+SP Risk Ratio Weight Risk Ratio<br />
1 Total Failure (P. falciparum) Day 28 PCR adjusted<br />
n/N n/N M-H,Fixed,95% CI M-H,Fixed,95% CI<br />
Mukhtar 2005 SDN 7/79 7/72 40.2 % 0.91 [ 0.34, 2.47 ]<br />
Karunajeewa 2007 PNG 3/96 11/98 59.8 % 0.28 [ 0.08, 0.97 ]<br />
Subtotal (95% CI) 175 170 100.0 % 0.53 [ 0.25, 1.13 ]<br />
Total events: 10 (AL6), 18 (AS+SP)<br />
Heterogeneity: Chi 2 = 2.16, df = 1 (P = 0.14); I 2 =54%<br />
Test <strong>for</strong> overall effect: Z = 1.64 (P = 0.10)<br />
2 Total Failure Day 28 PCR adjusted (trials with baseline differences included)<br />
Bousema 2004 KEN 1/73 11/142 22.0 % 0.18 [ 0.02, 1.34 ]<br />
Van den Broek 2004 ZAR 0/87 7/71 24.3 % 0.05 [ 0.00, 0.94 ]<br />
Mukhtar 2005 SDN 7/79 7/72 21.6 % 0.91 [ 0.34, 2.47 ]<br />
Karunajeewa 2007 PNG 3/96 11/98 32.1 % 0.28 [ 0.08, 0.97 ]<br />
Subtotal (95% CI) 335 383 100.0 % 0.34 [ 0.17, 0.66 ]<br />
Total events: 11 (AL6), 36 (AS+SP)<br />
Heterogeneity: Chi 2 = 5.86, df = 3 (P = 0.12); I 2 =49%<br />
Test <strong>for</strong> overall effect: Z = 3.21 (P = 0.0013)<br />
3 Total Failure Day 28 PCR adjusted (indeterminate PCR included as failures)<br />
Mukhtar 2005 SDN 7/79 7/72 36.3 % 0.91 [ 0.34, 2.47 ]<br />
Karunajeewa 2007 PNG 5/98 13/100 63.7 % 0.39 [ 0.15, 1.06 ]<br />
Subtotal (95% CI) 177 172 100.0 % 0.58 [ 0.29, 1.16 ]<br />
Total events: 12 (AL6), 20 (AS+SP)<br />
Heterogeneity: Chi 2 = 1.38, df = 1 (P = 0.24); I 2 =28%<br />
Test <strong>for</strong> overall effect: Z = 1.54 (P = 0.12)<br />
4 Total Failure Day 28 PCR adjusted (new infections included as successes)<br />
Mukhtar 2005 SDN 7/80 7/77 35.3 % 0.96 [ 0.35, 2.62 ]<br />
Karunajeewa 2007 PNG 5/113 13/112 64.7 % 0.38 [ 0.14, 1.03 ]<br />
Subtotal (95% CI) 193 189 100.0 % 0.59 [ 0.29, 1.17 ]<br />
Total events: 12 (AL6), 20 (AS+SP)<br />
Heterogeneity: Chi 2 = 1.66, df = 1 (P = 0.20); I 2 =40%<br />
Test <strong>for</strong> overall effect: Z = 1.52 (P = 0.13)<br />
5 Total Failure Day 28 PCR adjusted (losses to follow up included as failures)<br />
0.005 0.1 1 10 200<br />
Favours AL6 Favours AS+SP<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
(Continued ...)<br />
182