Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
Artemisinin-based combination therapy for ... - The Cochrane Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
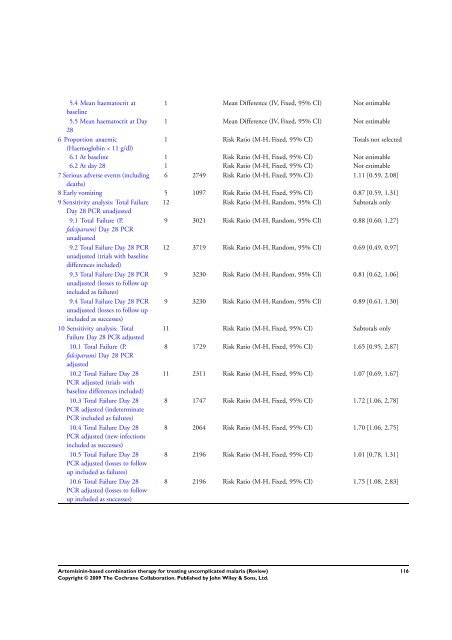
5.4 Mean haematocrit at<br />
baseline<br />
1 Mean Difference (IV, Fixed, 95% CI) Not estimable<br />
5.5 Mean haematocrit at Day<br />
28<br />
1 Mean Difference (IV, Fixed, 95% CI) Not estimable<br />
6 Proportion anaemic<br />
(Haemoglobin < 11 g/dl)<br />
1 Risk Ratio (M-H, Fixed, 95% CI) Totals not selected<br />
6.1 At baseline 1 Risk Ratio (M-H, Fixed, 95% CI) Not estimable<br />
6.2 At day 28 1 Risk Ratio (M-H, Fixed, 95% CI) Not estimable<br />
7 Serious adverse events (including<br />
deaths)<br />
6 2749 Risk Ratio (M-H, Fixed, 95% CI) 1.11 [0.59, 2.08]<br />
8 Early vomiting 5 1097 Risk Ratio (M-H, Fixed, 95% CI) 0.87 [0.59, 1.31]<br />
9 Sensitivity analysis: Total Failure<br />
Day 28 PCR unadjusted<br />
12 Risk Ratio (M-H, Random, 95% CI) Subtotals only<br />
9.1 Total Failure (P.<br />
falciparum) Day 28 PCR<br />
unadjusted<br />
9 3021 Risk Ratio (M-H, Random, 95% CI) 0.88 [0.60, 1.27]<br />
9.2 Total Failure Day 28 PCR<br />
unadjusted (trials with baseline<br />
differences included)<br />
12 3719 Risk Ratio (M-H, Random, 95% CI) 0.69 [0.49, 0.97]<br />
9.3 Total Failure Day 28 PCR<br />
unadjusted (losses to follow up<br />
included as failures)<br />
9 3230 Risk Ratio (M-H, Random, 95% CI) 0.81 [0.62, 1.06]<br />
9.4 Total Failure Day 28 PCR<br />
unadjusted (losses to follow up<br />
included as successes)<br />
9 3230 Risk Ratio (M-H, Random, 95% CI) 0.89 [0.61, 1.30]<br />
10 Sensitivity analysis: Total<br />
Failure Day 28 PCR adjusted<br />
11 Risk Ratio (M-H, Fixed, 95% CI) Subtotals only<br />
10.1 Total Failure (P.<br />
falciparum) Day 28 PCR<br />
adjusted<br />
8 1729 Risk Ratio (M-H, Fixed, 95% CI) 1.65 [0.95, 2.87]<br />
10.2 Total Failure Day 28<br />
PCR adjusted (trials with<br />
baseline differences included)<br />
11 2311 Risk Ratio (M-H, Fixed, 95% CI) 1.07 [0.69, 1.67]<br />
10.3 Total Failure Day 28<br />
PCR adjusted (indeterminate<br />
PCR included as failures)<br />
8 1747 Risk Ratio (M-H, Fixed, 95% CI) 1.72 [1.06, 2.78]<br />
10.4 Total Failure Day 28<br />
PCR adjusted (new infections<br />
included as successes)<br />
8 2064 Risk Ratio (M-H, Fixed, 95% CI) 1.70 [1.06, 2.75]<br />
10.5 Total Failure Day 28<br />
PCR adjusted (losses to follow<br />
up included as failures)<br />
8 2196 Risk Ratio (M-H, Fixed, 95% CI) 1.01 [0.78, 1.31]<br />
10.6 Total Failure Day 28<br />
PCR adjusted (losses to follow<br />
up included as successes)<br />
8 2196 Risk Ratio (M-H, Fixed, 95% CI) 1.75 [1.08, 2.83]<br />
<strong>Artemisinin</strong>-<strong>based</strong> <strong>combination</strong> <strong>therapy</strong> <strong>for</strong> treating uncomplicated malaria (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
116