Labelling Review row-Online
Labelling Review row-Online
Labelling Review row-Online
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Lambda Light Chain<br />
Clone SHL53 New!<br />
1 mL, 0.1 mL liquid NCL-L-LAM-578 P (HIER)<br />
Clone HP-6054<br />
1 mL, 0.1mL lyophilized NCL-LAM FPW<br />
Polyclonal<br />
1 mL lyophilized NCL-LAMp P (Enzyme) W<br />
The basic structure of an immunoglobulin molecule consists of two identical<br />
heavy chains, either �, �, �, � or �, and two identical light chains, either<br />
kappa or lambda. Any heavy chain can associate with either light chain but<br />
on any immunoglobulin molecule both light chains are of the same type. The<br />
ratio of kappa and lambda light chains varies between Ig classes and<br />
subclasses. In a polyclonal population the ratio of kappa to lambda bearing<br />
B cells is approximately 2:1, with individual B cells thought to express kappa<br />
or lambda light chains, never both. The majority of kappa and lambda chains<br />
are bound to heavy chain immunoglobulin, however in normal individuals<br />
low levels of free light chain are present in serum. The occurrence of a<br />
mixture of kappa and lambda chain expressing cells suggests a polyclonal<br />
population and a reactive or nonneoplastic proliferation of B cells.<br />
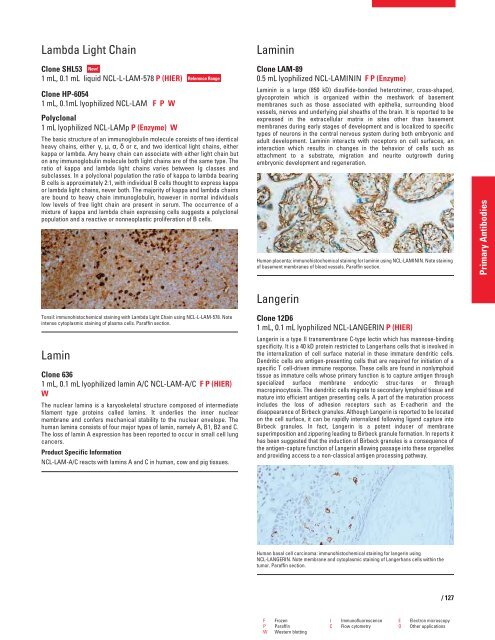
Tonsil: immunohistochemical staining with Lambda Light Chain using NCL-L-LAM-578. Note<br />
intense cytoplasmic staining of plasma cells. Paraffin section.<br />
Lamin<br />
Reference Range<br />
Clone 636<br />
1 mL, 0.1 mL lyophilized lamin A/C NCL-LAM-A/C F P (HIER)<br />
W<br />
The nuclear lamina is a karyoskeletal structure composed of intermediate<br />
filament type proteins called lamins. It underlies the inner nuclear<br />
membrane and confers mechanical stability to the nuclear envelope. The<br />
human lamina consists of four major types of lamin, namely A, B1, B2 and C.<br />
The loss of lamin A expression has been reported to occur in small cell lung<br />
cancers.<br />
Product Specific Information<br />
NCL-LAM-A/C reacts with lamins A and C in human, cow and pig tissues.<br />
Laminin<br />
Clone LAM-89<br />
0.5 mL lyophilized NCL-LAMININ F P (Enzyme)<br />
Laminin is a large (850 kD) disulfide-bonded heterotrimer, cross-shaped,<br />
glycoprotein which is organized within the meshwork of basement<br />
membranes such as those associated with epithelia, surrounding blood<br />
vessels, nerves and underlying pial sheaths of the brain. It is reported to be<br />
expressed in the extracellular matrix in sites other than basement<br />
membranes during early stages of development and is localized to specific<br />
types of neurons in the central nervous system during both embryonic and<br />
adult development. Laminin interacts with receptors on cell surfaces, an<br />
interaction which results in changes in the behavior of cells such as<br />
attachment to a substrate, migration and neurite outg<strong>row</strong>th during<br />
embryonic development and regeneration.<br />
Human placenta: immunohistochemical staining for laminin using NCL-LAMININ. Note staining<br />
of basement membranes of blood vessels. Paraffin section.<br />
Langerin<br />
Clone 12D6<br />
1 mL, 0.1 mL lyophilized NCL-LANGERIN P (HIER)<br />
Langerin is a type II transmembrane C-type lectin which has mannose-binding<br />
specificity. It is a 40 kD protein restricted to Langerhans cells that is involved in<br />
the internalization of cell surface material in these immature dendritic cells.<br />
Dendritic cells are antigen-presenting cells that are required for initiation of a<br />
specific T cell-driven immune response. These cells are found in nonlymphoid<br />
tissue as immature cells whose primary function is to capture antigen through<br />
specialized surface membrane endocytic struc-tures or through<br />
macropinocytosis. The dendritic cells migrate to secondary lymphoid tissue and<br />
mature into efficient antigen presenting cells. A part of the maturation process<br />
includes the loss of adhesion receptors such as E-cadherin and the<br />
disappearance of Birbeck granules. Although Langerin is reported to be located<br />
on the cell surface, it can be rapidly internalized following ligand capture into<br />
Birbeck granules. In fact, Langerin is a potent inducer of membrane<br />
superimposition and zippering leading to Birbeck granule formation. In reports it<br />
has been suggested that the induction of Birbeck granules is a consequence of<br />
the antigen-capture function of Langerin allowing passage into these organelles<br />
and providing access to a non-classical antigen processing pathway.<br />
Human basal cell carcinoma: immunohistochemical staining for langerin using<br />
NCL-LANGERIN. Note membrane and cytoplasmic staining of Langerhans cells within the<br />
tumor. Paraffin section.<br />
F Frozen I Immunofluorescence E Electron microscopy<br />
P Paraffin C Flow cytometry O Other applications<br />
W Western blotting<br />
/ 127<br />
Primary Antibodies