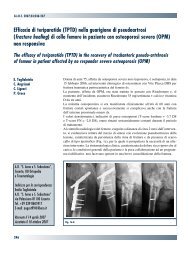
30845 Suppl Giot.pdf - Giornale Italiano di Ortopedia e Traumatologia
30845 Suppl Giot.pdf - Giornale Italiano di Ortopedia e Traumatologia
30845 Suppl Giot.pdf - Giornale Italiano di Ortopedia e Traumatologia
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
G.I.O.T. 2010;36(suppl. 1):S60-S62<br />
Bacterial biofilms: the ultimate cause of implant-associated infections<br />
Film batterico: causa primitiva delle infezioni negli impianti protesici<br />
E. Meyle, G. M. Hänsch<br />
Bacterial infections occurring after osteosynthesis or implantation<br />
of prostheses are serious complications in orthopae<strong>di</strong>c surgery.<br />
In the majority of cases, an antibiotic therapy is not successful.<br />
Infections therefore persist and can give rise to a progressive,<br />
destructive inflammatory process with massive tissue injury and<br />
osteolysis, an entity known as implant-associated posttraumatic<br />
osteomyelitis (for review see 1-5 ). In most cases the removal of the<br />
implant, which might require extensive debridement and intricate,<br />
costly reconstructive surgery, is the only option. Therefore, in the<br />
recent years, considerable effort was put into unravelling the pathogenesis<br />
of implant-associated osteomyelitis as a prerequisite for an<br />
effective and eventually also causal therapy.<br />
It became apparent that a specialised life-form of bacteria, the<br />
so-called “biofilm” was responsible for the persistent infection.<br />
Biofilms are defined as bacterial communities, where the bacteria<br />
are embedded in an extracellular polymeric substance, morphologically<br />
apparent as slime or film. In the case of implant-infection, the<br />
biofilm is attached rather firmly to the implant. The formation of<br />
the film is genetically programmed, with production of slime as a<br />
hallmark (reviewed in 6-8 ).<br />
Biofilm formation has been demonstrated for numerous bacteria<br />
species. In implant-associated infection, staphylococci are prevalent,<br />
and experimental data show that staphylococci rea<strong>di</strong>ly attach<br />
to metal surfaces, especially in the presence of serum or plasma,<br />
and form biofilms there 9-11 . In vitro data imply that rough surfaces,<br />
as they are generated by handling for example – as shown in<br />
Fig. 1A – by clipping a wire favours the attachment of bacteria and<br />
hence biofilm formation (Fig. 1A).<br />
The slimy matrix protects the bacteria against unfavourable<br />
environmental con<strong>di</strong>tions, among others against antibiotics or<br />
biocides.<br />
How the immune system deals with biofilm infection is not yet<br />
well understood. Most of our knowledge on bacteria-host defence<br />
interactions is derived from studying single, free swimming “planktonic”<br />
bacteria. Whether similar interactions occur also in biofilm<br />
infection is unknown. Moreover, the link between biofilm infection<br />
and the destructive inflammatory reaction is not yet understood,<br />
nor the pathomechanism of the ensuing osteolysis, that is a serious<br />
complication of implant-associated osteomyelitis.<br />
To gain insight into the host defence against biofilm infections, the<br />
main object of our experimental research is focussed on systemic<br />
and local immune reaction in patients with implant-associated<br />
osteomyelitis. In that context, we assessed interactions between<br />
cells of the host defence with biofilms in vitro.<br />
Institut für Immunologie der Universität Heidelberg, Germany<br />
S60<br />
In the following, we will briefly summarise our fin<strong>di</strong>ngs:<br />
1. Implant-associated osteomyelitis is a localised infection.<br />
Accor<strong>di</strong>ngly, systemic markers of infections, inclu<strong>di</strong>ng fever<br />
or increased leukocyte counts are not regularly seen, but<br />
enhanced serum concentration of C-reactive protein (CRP)<br />
are found in approximately 85 % of the patients (our own data<br />
comprising 92 patients). An activation of phagocytic cells was<br />
also seen in the majority of patients, particularly of neutrophils,<br />
the so-called “first-line defence” against bacterial infection.<br />
Moreover, neutrophils, and to a lesser extent, T-lymphocytes,<br />
infiltrate the infected site, and can be recovered during surgery<br />
for functional analysis ex vivo. We found that these cells were<br />
highly activated, and “primed” for bactericidal and cytotoxic<br />
activity 12-15 (Fig. 1C; 1D).<br />
2. The presence of neutrophils at the site of infection leads to<br />
the question whether these cells would be able to destroy biofilms.<br />
In order to analyse the interactions between PMN and<br />
biofilms of the staphylococci species we generated biofilms in<br />
vitro. Afterwards neutrophils from healthy donors were added<br />
and the effects were observed by means of time-lapse video<br />
microscopy. We saw that PMN migrated towards and across<br />
biofilms, and cleared areas of biofilm as they moved along.<br />
By laser scan microscopy and cytofluorometry we could detect<br />
and measure uptake of the bacteria by phagocytosis, similar to<br />
the process known for free swimming “planktonic” bacteria.<br />
After phagocytosis, the neutrophils <strong>di</strong>ed by a process known as<br />
”programmed cell death” (apoptosis) 16 (Fig. 1E-H).<br />
3. Imme<strong>di</strong>ately after contact with the biofilm, the neutrophils also<br />
released lactoferrin, that in ad<strong>di</strong>tion to its bactericidal activity<br />
is also known to inhibit biofilm formation what we could confirm<br />
in our experiments 17 (Fig. 1).<br />
In Summary, our data in<strong>di</strong>cate that biofilm infections are rea<strong>di</strong>ly<br />
recognised by the immune system; Moreover, the appropriate<br />
immune cells infiltrate the infected site and are activated there; and<br />
in ad<strong>di</strong>tion the biofilms are not inherently protected against the<br />
defence by neutrophils – at least in vitro.<br />
Whether biofilms are attacked and cleared by PMN in vivo is <strong>di</strong>fficult<br />
to prove, because a localised immune defence is likely to<br />
occur unnoticed by the patients. Only unsuccessful attempts of the<br />
host defence become apparent, particularly in late stages, when the<br />
tissue damage is already in an advanced state and the implant is<br />
loosening.<br />
So the questions arise why in those patients the host defence is<br />
insufficient and how the infection is linked to the tissue damage.<br />
With regard to the first question, the classical para<strong>di</strong>gm for infection<br />
“too little – too late” might hold true also for biofilm infection.